Samsung NeuroLogica has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Auto Lung Nodule Detection (ALND) tool, an artificial intelligence (AI)-assisted detection program for locating pulmonary nodules.
The tool is specifically designed to aid physicians in reviewing posteroanterior chest radiographs in adults for nodules 10 mm to 30 mm in size. The program is part of S-Station, the operating software installed on Samsung's digital x-ray imaging systems and includes an autorun option that automatically performs nodule detection after chest x-ray imaging.
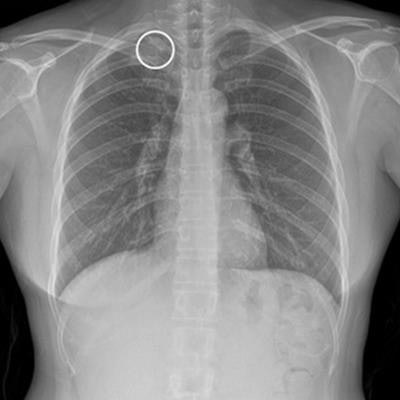
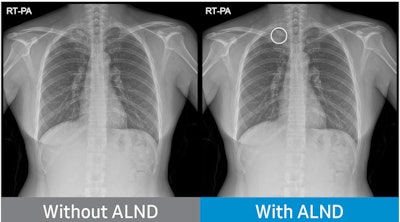 Chest radiograph without Auto Lung Nodule Detection (ALND) and chest radiograph with lung nodule marked. Image courtesy of Neuologica.
Chest radiograph without Auto Lung Nodule Detection (ALND) and chest radiograph with lung nodule marked. Image courtesy of Neuologica.In international clinical trials, ALND achieved a sensitivity of at least 80% identifying lung cancer in 600 chest x-rays, the company said.