Toronto-based Conavi Medical has received clearance from the U.S. Food and Drug Administration (FDA) for its next-generation hybrid intravascular imaging system.
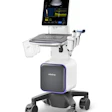
The system integrates intravascular ultrasound (IVUS) and optical coherence tomography (OCT) into a single platform and enables simultaneous, co-registered imaging in a single pullback. The system allows physicians to visualize both deep vessel structures and high-resolution surface detail in real time, according to the company. It also includes lesion and stent analysis tools that incorporate data from both modalities, the firm said.
Conavi expects to begin a limited U.S. market release in select centers in the second half of 2026.