Researchers have developed a quantitative MRI-CT fat phantom that is designed to harmonize CT-derived myosteatosis measurements with MRI-derived proton density fat fraction (PDFF), and across imaging protocols, platforms, and vendors, according to a poster to be presented May 12 at the ISMRM meeting in Cape Town, South Africa.
Development of a reference standard targeting myosteatosis -- abnormal fat accumulation in skeletal muscle -- on both CT and MRI supports opportunistic screening for this prognostic indicator that has been shown to be a strong predictor of reduced overall survival in multiple populations, including patients with a variety of cancers.
Myosteatosis is also related to increased frailty and decreased muscle and mobility function in aging, noted a group led by Calimetrix and University of Wisconsin-Madison researchers including first author, David Rutkowski, PhD, presenting author Jean Brittain, PhD, and Scott Reeder, MD, PhD.
"While access to quantitative MRI methods to measure PDFF remains limited, CT is ubiquitously available and used more frequently than MRI in clinical abdominal imaging," Brittain told AuntMinnie in an email. There is growing interest in quantifying myosteatosis, with broad potential applications in clinical care and clinical trials, according to the group.
Phantom extended
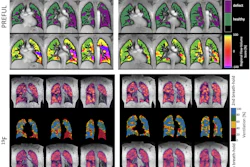
Extending work presented in 2025, the group highlighted data confirming the phantom's performance, as assessed on a 3-tesla (3T) clinical MRI system (Signa Premier, GE HealthCare) using a commercial chemical shift encoded MRI (CSE-MRI) method to quantify PDFF using IDEAL IQ volumetric fat-fraction mapping; and a 64-slice multidetector CT scanner (Discovery CT750 HD, GE HealthCare).
The phantom formulation was based on a retrospective analysis led by Adam Kuchnia, PhD, of 50 asymptomatic adults who underwent CT colonographic screening between February 2013 and June 2014. Patients underwent both CT and MR imaging on the same day to measure CT number and PDFF values in paraspinal muscle.
Patient MR data were acquired with a 1.5T system (Signa HDxt, GE HealthCare) using quantitative CSE-MRI (44 × 44 cm FOV, 256 × 160 matrix, 8-mm slice thickness, 32 slices, 5° flip angle, ± 125-kHz receiver bandwidth; TR, 13.6 ms; and six echoes [initial TE, 1.20 ms; ΔTE, 1.98 ms]).
Twelve emulsions were formulated with PDFF targets and CT number targets corresponding to the in-vivo muscle data fit from the retrospective analysis. Ingredient concentrations were iteratively modulated and tested for each target value until a formulation was achieved within ±2% absolute PDFF and a CT number with overall bias < ±10 HU with 95% confidence relative to the known in vivo muscle CT number at a given PDFF value. Phantom results were compared.
Images were analyzed by placing regions of interest in the cross-section of each vial, the group explained.
The results revealed the relationship of CT number to PDFF in paraspinal muscle tissue to be CT#(HU) = –2.25* [MRI PDFF(%)] + 65.4, according to the group. In the phantom vials, the CT# to PDFF relationship was characterized as CT#(HU) = –2.27* [PDFF (%)] + 63.6. The CT to MRI relationship of the phantom formulation closely mimicked that of in-vivo muscle tissue, Brittain noted.
Last year, the group introduced the general CT–MRI fat phantom that mimics different concentrations of liver fat on both CT and MRI. That phantom is now commercially available through Calimetrix. However, muscle has different CT attenuation behavior compared to the liver and therefore additional development work was required, Brittain explained.
Potential QA tool
Overall, the work is important because of the growing prevalence chronic diseases. Over 90 million Americans are imaged using CT every year for a wide range of clinical indications, and the liver and skeletal muscles are visualized in a large proportion of these studies, Brittain said.
"A multimodality imaging phantom that mimics the quantitative PDFF and CT number measurements of myosteatotic paraspinal tissue was successfully developed," the group noted. "This phantom has the potential to serve as an essential tool for quality assurance and harmonization of MRI and CT quantitative imaging biomarkers of myosteatosis."
Both projects were funded by Small Business Innovation Research (SBIR) grants from the National Institute of Standards and Technology (NIST).
Check out AuntMinnie's full coverage of ISMRM 2026 on our ShowCast.