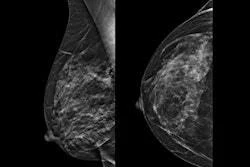
Lunit has received clearance from the U.S. Food and Drug Administration (FDA) for version 1.2 of its 3D mammography AI algorithm. The announcement was made at the Society of Breast Imaging (SBI) Annual Symposium in Seattle.
The updated algorithm introduces current-prior comparison and multiple operating thresholds to its Insight breast imaging platform. It provides AI findings for the current exam and up to two prior exams, and offers three selectable thresholds to support different sensitivity and specificity trade-offs, the company said. This version also integrates Volpara Scorecard volumetric breast density measures alongside AI scores.
Lunit also highlighted a milestone at SBI 2026, noting that it has expanded to more than 330 screening sites across the Americas over the past year -- equal to supporting approximately one million screening mammograms annually, the firm said.