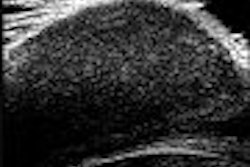
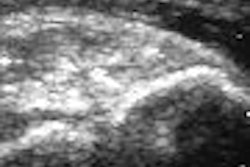
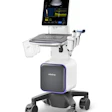
Siemens Medical Solutions of Malvern, PA, has starting shipping Acuson P10, a handheld diagnostic ultrasound system that's slightly larger than a personal digital assistant (PDA).
The device, which has received U.S. Food and Drug Administration 510(k) clearance, weighs 1.6 lb and fits into a lab coat pocket. It is intended for complementary initial diagnostic care and triage, particularly in cardiology, emergency care, and obstetrics.
Acuson P10 is designed for physicians and medical personnel in a number of environments, including intensive care units (ICUs), ambulances, and medevac helicopters, to detect conditions that may be clinically significant but that previously required more intensive testing.
The company first introduced the scanner as a work-in-progress at the 2006 RSNA meeting.
By AuntMinnie.com staff writers
October 2, 2007
Related Reading
Siemens boosts Japan activities, October 1, 2007
Siemens extends date for Dade Behring deal, September 27, 2007
Siemens launches European e-health project, September 21, 2007
Siemens scores U.S. antitrust OK for Dade Behring deal, September 19, 2007
Siemens to buy lab giant Dade Behring for $7 billion, July 25, 2007
Copyright © 2007 AuntMinnie.com