CHICAGO - A pair of new CT scanners is pacing the RSNA 2011 product introductions from Siemens Healthcare. The company is also promoting Biograph PET/CT and its new Acuson S1000 ultrasound system, and is demonstrating new product launches in healthcare informatics, digital radiography, and interventional radiology.
CT
The new CT scanners include Somatom Definition Edge, a single-source version of the company's flagship dual-source Somatom Flash system, and Somatom Perspective, an economical system designed to provide 128-slice scanning at a lower price point.
With Edge, Siemens wanted to create a new product that included all of the functionality of Flash, but without the dual-source design, which contributes to a higher price for the top-of-the-line system due to the double detector and x-ray tube arrays. Edge will carry a price point that is 30% to 40% lower than Flash, according to the company.
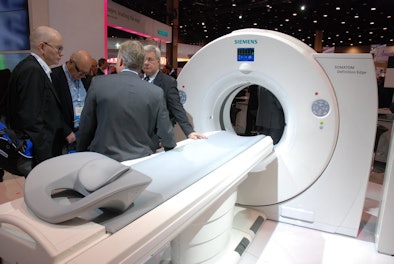 |
| Siemens is introducing its new Somatom Definition Edge CT scanner. |
Edge sports a 0.28-sec gantry rotation speed, with 23 cm of patient coverage -- exactly half that of Flash. This scanner's speed makes it possible to acquire a thorax-abdominal scan in about two seconds, and it also reduces the need to have patients hold their breath during scans. The scanner acquires images at 64 slices and reconstructs at 128 slices due to the company's z-Sharp technology.
Siemens is also touting a dual-energy mode in which two scans are performed in succession with different energy levels, with each scan using half the dose of a normal scanning mode. Because the tube's voltage is fixed during the mode, users can employ dose-reduction tools such as CareDose4D.
Meanwhile, Somatom Perspective delivers 128-slice scanning at a more economical price point, without many of the premium features found on Edge (like Edge, it acquires images at 64 slices and reconstructs at 128). The scanner has a 0.48-sec rotation speed; it also includes iTRIM (iterative temporal resolution improvement method), an algorithm designed to improve temporal resolution for cardiac studies. With iTRIM, Perspective has a temporal resolution of 195 msec, which is the equivalent of a scanner with a much faster gantry rotation speed, according to the company.
Both Perspective and Edge are pending 510(k) clearance with the U.S. Food and Drug Administration (FDA), and are being shown as works-in-progress. Commercial shipments are expected in the middle of 2012.
In addition to the new scanners, Siemens is launching a new detector design, called Stellar, at this year's RSNA 2011 meeting. Stellar detectors are used in the new Edge scanner and will also be available for the Flash system.
In designing Stellar, Siemens integrated all of the components into the electronics board, resulting in a design it calls TrueSignal that reduces noise by 30% and produces better signal-to-noise ratios. The design is especially useful when processing images with iterative reconstruction techniques -- in fact, Siemens executives said that Stellar was designed specifically to support iterative reconstruction.
Users of a scanner equipped with Stellar detectors can elect to either scan at the same radiation dose but with higher image quality, or scan at a lower radiation dose with equivalent image quality. Scanners using the detectors have a spatial resolution of 0.33 mm.
The company is awaiting 510(k) clearance for Stellar, and upgrades are expected to be offered to the installed base of Flash systems in the middle of 2012.
In a bit of fortuitous timing, Siemens' sinogram-affirmed iterative reconstruction (SAFIRE) algorithm received FDA clearance just days before the opening of RSNA 2011. SAFIRE includes the image reconstruction software and hardware, and yields CT radiation dose reduction of up to 60% compared with previous filtered back-projection techniques, according to the company.
Finally, Siemens is discussing eMode, a new software-based scanning technique that optimizes scanning parameters to reduce wear and tear on the system and prolong its useful life. For example, most users might not need to operate the system at the highest pitch and mA settings; reducing parameters to 90% of the highest output would have little impact on most clinical applications. Siemens is also able to track scan usage with eMode, and the firm can offer customers who use it extensively a lower rate for service costs.
Ultrasound
Siemens is launching its new Acuson S1000 ultrasound system in Chicago as the newest addition to its portfolio. The Acuson S1000 has the same hardware and architecture as the Acuson S2000 and S3000 models and can be upgraded with the higher-end features.
The Acuson S1000 also offers 20 transducers and 26 applications and has received 510(k) clearance. Siemens plans to begin shipments of the device in early 2012, targeting community hospitals, large healthcare systems, imaging centers, and physician practices.
 |
| Siemens' new Acuson S1000 ultrasound system. |
Siemens is also showing its new Acuson S3000 premium ultrasound system, which features eSie Fusion imaging and multimodality (CT and MRI) review for additional clinical and spatial information in the analysis of complex pathology and/or when performing interventional procedures, such as biopsies.
eSie Fusion imaging is designed for the automatic fusion of 3D CT with real-time ultrasound in seconds, rather than in minutes with manual registration techniques, and to enhance workflow during MR volume registration.
The Acuson S3000 is pending 510(k) clearance from the FDA, while eSie Fusion imaging currently is a work-in-progress.
Siemens is also introducing its new 8C3 HD transducer designed for obstetrics and pediatric applications. The transducer features a small footprint for enhanced surface contact and scanning performance. A 50% larger field-of-view enables the display of a full third-trimester fetal head, improving measurements and anatomic visualization.
The company is also spotlighting the 3.0 release of Acuson S2000, which adds two transducers to its existing line. The 6C1 HD (high-density) abdominal imaging transducer and the V7M TEE (transesophageal echocardiogram) pediatric cardiology transducer feature sensitivity enhancements to Siemens Cadence contrast pulse sequencing technology.
Healthcare informatics
In healthcare informatics, Siemens is discussing the latest release of its syngo.via software, VA11. Siemens has doubled the number of syngo.via apps currently available, which now stands at 62, and the company has also added apps to support cardiac cath labs. The company is also targeting the market for mobile computing, making syngo.via apps available in areas such as Apple's iTunes store. Siemens is also showing a version of syngo.via running in a cloud-based configuration, but the company has not set a release date yet for that functionality.
Siemens has also made progress integrating syngo applications for radiology and cardiology to enable physicians using either software to more easily view images from the other in a package Siemens is calling Cardiovascular Workplace. Users of syngo.via VA11 and the newest version of the company's cardiology application, syngo Dynamics V9.5, can now operate from a single worklist that displays data from both, so that radiology images can be viewed on the same display as cardiac measurements and other information. The integration also supports the creation of a single patient report.
For Siemens' syngo Workflow application, the company has added an integrated dose module, as well as the capability for radiologists to sign off on reports from mobile devices. The newest version, V32, also is now cloud-based, so users no longer have to buy separate hardware. V32 will begin shipping in early 2012.
On the company's syngo.plaza PACS software suite, Siemens is demonstrating version VA20C, which will begin shipping in December. New features include faster image loading speeds, support for the iPad, and support for Citrix-based installation.
Finally, as a demonstration project, Siemens is showing a touchless display controller interface based on Microsoft's Kinect video game controller. The interface enables a user to control a large-format display without touching any hardware, which could be a useful feature for a surgeon in the operating room, for example. Siemens did not specify whether it actually planned to release the interface as a commercial product.
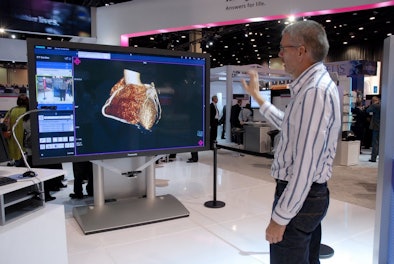 |
| The interface based on Microsoft's Kinect allows users to control a display without touching any hardware. |
Interventional radiology, x-ray
In its angiography line, Siemens is highlighting a new DynaCT 360 application for its Artis zeego angiography system. The system now performs a 360º spin in six seconds, as opposed to a 200º rotation in eight seconds for syngo DynaCT on the company's older systems. The ability should prove useful with imaging patients who aren't able to hold their breath for long periods, according to the company. Artis zeego with syngo DynaCT 360 will begin shipping in the summer of 2012.
Siemens is also making its Combined Applications to Reduce Exposure (CARE) dose reduction applications as standard features on all systems in the Artis zee family, meaning that users will no longer have to pay for the features as options. The company said it is about halfway through upgrading all the systems in its installed base.
The company is also showing embolization guidance, in which an MRI scan is used as the basis for angiography guidance during interventional procedures through torturous vessels, such as embolizing uterine fibroids. Siemens believes the technique will help reduce radiation dose.
Finally, Siemens is demonstrating what it calls Artis zee Cockpit Solution, in which video from multiple sources can be displayed on a single console in the angiography suite. The feature is designed to make it easier to manage the multiple imaging modalities that are now being used during interventional procedures. The feature is currently shipping.
On the radiography side, Siemens is introducing a new digital radiography (DR) system, called Multix Fusion, at RSNA 2011. The system is designed for small and medium-sized hospitals and is more affordable than earlier DR models in the Siemens product line, but also includes high-end features, according to the company.
 Siemens' new Multix Fusion DR system.
Siemens' new Multix Fusion DR system.
Multix Fusion uses a ceiling-mounted x-ray tube and also includes a wall bucky stand that was adapted from the company's high-end Ysio system. The system's tube automatically synchronizes itself when the detector is adjusted, enabling exams to be performed more quickly. Also, the unit's 1.8-meter-long telescoping column enables users to pull the tube down directly above the floor even in rooms with high ceilings, which can be helpful in positioning patients, such as for foot exams.
Other features include a patient table with a 660-lb capacity, installation in less than 20 square meters, and special service contracts that can be configured for the needs of individual customers. Multix Fusion is not currently shipping and is being shown as a work-in-progress.
Siemens is also highlighting Luminos Agile, a dual-use system for both radiography and fluoroscopy that's based on a flat-panel 17 x 17-inch digital detector. Siemens believes the design produces images that are up to 116% larger and have better patient coverage than a conventional 13-inch image intensifier, which reduces the need to reposition patients and thus reduces overall fluoroscopy imaging time. Siemens announced the first Luminos Agile installation this month.
Other x-ray highlights at RSNA 2011 for Siemens include the Ysio DR system, Mobilett Mira mobile DR unit with a wireless detector, and Arcadis Avantic mobile C-arm.
MRI
A year after its global introduction, Siemens' integrated PET/MRI system, the Biograph mMR, continues to provide simultaneous MR and PET imaging at research institutions and healthcare facilities. More than 20 Biograph mMR units have been ordered by university hospitals, research sites, and private practices, with approximately 15 systems installed worldwide.
Dr. Christoph Zindel, Siemens' vice president of MR, noted that the hybrid technology's applications in oncology, neurology, and cardiology have helped in the early detection of tumors and other maladies to assess treatment and alter therapies when necessary.
Siemens is also showcasing three new Dot (day optimizing throughput) software products to support image quality, workflow, and productivity. The Breast Dot Engine, Spine Dot Engine, and Large Joint Engine are all works-in-progress that are designed for additional clinical indications on the Magnetom Aera 1.5T and Magnetom Skyra 3T.
The Dot technology is designed to increase productivity by offering exam personalization, user guidance, and workflow automation. The company expects the Dot engines to be available around mid-2012.
Siemens also is expanding its Tim (total imaging matrix) integrated coil technology with TimTX TrueShape, a work-in-progress for parallel transmit imaging. Zindel said TimTX TrueShape is designed to selectively "excite" areas, such as the prostate, pancreas, or spine, and ignore surrounding structures to accentuate a particular region-of-interest.
Siemens is also adding its first parallel transmit application, syngo ZOOMit, to its MR product line. ZOOMit would allow a technologist to zoom in an MR image and accelerate the scan. This new technology and application will be fully integrated into the Magnetom Skyra 3T MRI.
Molecular imaging
Siemens is unveiling a new version of its Biograph PET/CT system to improve the quantification of standardized uptake values (SUVs) for detecting tumors and the subsequent evaluation of treatment efficacy.
 Siemens' Biograph PET/CT system.
Siemens' Biograph PET/CT system.
Rather than the gantry independently moving the patient into the PET/CT bore, the gantry and its supporting base travel together into the scanner to keep a patient parallel and avoid a downward angle the further the gantry travels. The enhanced stability allows for more precise PET/CT hybrid image quality.
Siemens is also adding Volumetric Resolution to enhance SUV calculations and OptisoHD (optimum isotropic high-definition resolution) to improve image resolution from the conventional standard of 135 mm3 to 87 mm3. Both features are currently available on the Biograph PET/CT.
In addition, QuantiQC will provide daily calibration for Biograph PET/CT, while Siemens' Molecular and Anatomical Recognition Technologies (SMART) software will enhance the alignment of PET and CT images in applications such as brain and heart imaging.
Siemens is also highlighting its Symbia line of SPECT and SPECT/CT scanners, as well as new areas in imaging biomarker production and distribution. Symbia features a range of specialty diagnostic tools, such as IQ-SPECT and automated productivity features.
Symbia IQ-SPECT is designed to reduce dose and imaging time in routine scans. Diagnostic SPECT/CT, including calcium scoring, uses half of a typical technetium dose and reduces acquisition time from approximately 20 minutes to less than five minutes with the addition of IQ-SPECT.
Refurbished systems
Siemens is also launching its new Ecoline refurbished product line at RSNA 2011 to provide scalable systems with a variety of refurbishment options for x-ray, angiography, CT, molecular imaging (SPECT/CT, PET, PET/CT), MRI, ultrasound, and oncology.
A 12-month warranty accompanies all devices.
Siemens' refurbishment process is intended to reduce annual CO2 emissions by as much as 20,000 tons, and the company's refurbished systems have an average material reuse rate of 98%.