CHICAGO - At the top of the agenda in the RSNA 2011 booth of GE Healthcare is a new laser-guided, robotic-controlled interventional angiography system that was unveiled with fanfare at this week's meeting. Also featured are patient-controlled environments for mammography and MRI, a new mobile x-ray system, and a new entry in the vendor's PET/CT product line.
Interventional radiology
GE made a major splash on Sunday afternoon with the unveiling of Discovery IGS 730, the company's new flagship angiography system. The system departs from traditional angiography instrumentation, in which the imaging gantry is mounted on either the floor or ceiling, in favor of a new design that employs a mobile wheel-mounted, self-powered stand that's directed by a precision laser-guidance system, according to the company.
GE developed the new design to address concerns about both floor- and ceiling-mounted angiography systems in terms of patient access and siting. The unit features a robotic guidance system at the foot of the gantry that receives GPS coordinates from sensors in the room. The system can be programmed to move into position, then park and retract both its wheels and cables for imaging. Cable sweepers in the bottom of the stand prevent the system from running over cables, while sensors in the base stop the system if it encounters an object.
Discovery IGS 730 is a single-plane system that also features a new C-arm design GE is calling WideBore 3D. The design is based on the company's Innova angiography line, but with an expanded C-arm that supports imaging with steeper angles, better 3D spins, and heavier patients. The new system also features Innova's imaging chain and a 30 x 30-cm digital detector, as well as 20 advanced imaging applications. Discovery IGS 730 is pending U.S. Food and Drug Administration (FDA) clearance and is being shown as a work-in-progress at this week's show.
 |
| Discovery IGS 730 features a new laser-guided robotic-controlled positioner. |
GE is also launching a new biplane angiography system in the Innova line, Innova IGS 630. The system uses a traditional gantry stand, but it features a new 3D imaging technology called Innova CT HD as an option. GE believes that CT HD provides higher-quality 3D imaging than previously available, without increasing radiation dose, by using high-frame-rate acquisition and an advanced scatter reduction algorithm. CT HD is well-suited for 3D imaging of areas such as bones and soft tissues, and it includes a high-resolution mode for visualizing small objects such as intracranial stents.
On the application side, new tools that GE is touting include TrackVision for planning and guiding needle procedures, and AngioViz for digital subtraction angiography (DSA), which enables vascular flow to be viewed on a single image.
GE is also discussing enhancements to its Mobile Hybrid OR system, which combines a mobile C-arm, a mobile table, and an integrated video system, including wireless data transfer to PACS and wireless remote service. In addition, GE is highlighting its dose reduction initiatives, which include user training activities and an iPad app to help operating room staff operate the system as efficiently as possible, according to the vendor.
MRI
The highlight of the MRI section of GE's booth isn't a scanner, but an idea: that patients have a right to a scanning environment that's less intimidating than what they currently experience. To soften radiology's sometimes-rough edges, GE has launched the MR Patient Experience Suite.
Before scanning begins, patients entering the suite are given an iPad that enables them to control the environment, with 16 themes that include different lighting and ambient sound options -- a single touch to the iPad changes the room environment instantly. An LCD monitor on the ceiling displays nature scenes specifically chosen to be soothing for patients. Finally, all cables and supplies are stored behind wall cabinets to minimize the "clinical" feel of the scanning room.
GE customers who are using the company's w-series MRI scanners can order the suites through GE's partner, PDC Facilities, and the companies plan to begin the first installations of the suite in January 2012.
In more traditional MRI news, GE is highlighting a new algorithm called MAVRIC SL, designed to make it easier for imaging facilities to conduct MRI scans on patients with metallic implants. Traditionally, scans of patients with such implants suffer from severe artifacts, and the problem is growing as the number of individuals undergoing implant procedures increases: GE estimates that between 1990 and 2002, the number of arthroplasty procedures in the U.S. increased by 62% for total hip replacements and by 195% for total knee replacements.
MAVRIC SL attacks the problem by combining image acquisition and reconstruction techniques developed with Stanford University and the Hospital for Special Surgery. GE expects that the protocol will reduce artifacts and make it easier for doctors to visualize tissue next to metal implants, and to more easily correct complications that might result from surgical implant procedures. The technique is pending FDA clearance and will be available on GE 1.5-tesla and 3-tesla scanners.
On the company's Optima MR450w scanner, GE is highlighting ongoing work in MR elastography applications, as well as Ideal IQ, which derives images that enable clinicians to determine a patient's fat content in cases of liver disease. GE is also highlighting its GEM Suite of flexible coils, which can be tailored for whatever anatomical part is being imaged.
Finally, the company is promoting its Optima 430s extremity scanner, which the company said has been very popular in Europe. GE is planning to roll out a mobile version of the system for the U.S.
X-ray
Optima XR220amx is a new mobile x-ray system that GE is launching at this week's RSNA 2011 meeting. As an option, the system can be configured with GE's FlashPad wireless digital radiography (DR) detector, which was introduced at the 2010 RSNA show.
 GE's FlashPad is now available with several new systems.
GE's FlashPad is now available with several new systems.
Optima XR220amx is designed to bring the image quality of DR to locations like the emergency room, intensive care unit, and other clinical settings. The system also has ergonomic improvements for better patient transportation and positioning, such as touchscreen controls, immediate startup capability, and more image storage.
GE also announced that FlashPad, which features ultrawideband wireless connectivity, is now available in several new radiography and radiography/fluoroscopy systems, including GE's Discovery XR656 fixed DR system.
In bone densitometry, GE is launching CoreScan, a new feature for its iDXA dual-energy x-ray absorptiometry (DEXA) systems that quantifies visceral adipose tissue (VAT), a type of body fat that is linked to diabetes and cardiac disease. The scans can help clinicians differentiate VAT from subcutaneous fat, which is believed to be less of a health risk.
Also in women's health, GE is promoting its SenoBright contrast-enhanced spectral mammography (CESM) mode for full-field digital mammography. SenoBright was first introduced at the 2010 RSNA show, and it received FDA clearance in October 2011. Also being highlighted in the company's booth is the molecular breast imaging system that received FDA clearance last year.
Finally, GE is demonstrating a mammography version of its Patient Experience Suite concept, which tries to make the imaging experience less stressful for patients by adding mood lighting, soothing music, and even scents to the imaging suite. As with the MRI suite, patients can control the environment they want before starting an imaging procedure.
CT
 Veo received FDA clearance in August.
Veo received FDA clearance in August.
Radiation dose has become a major issue in radiology over the past several years, and it's a highlight in the CT section of GE's booth as well. At the RSNA 2011 show, the company announced that it has made an additional $300 million R&D investment in developing technologies for reducing and tracking radiation dose. The investment comes on top of $500 million in low-dose research the company has made since 2000, a figure that encompasses CT, x-ray, and informatics areas.
An example of this investment is the DoseWatch software that GE launched in October at the annual congress of the French Radiology Society (SFR), les Journées Françaises de Radiologie (JFR). DoseWatch meets the growing need for better tracking and reporting of the radiation doses that patients receive, and it can track dose from multiple vendors and imaging modalities. The software is offered in a server-based configuration, and GE has begun selling licenses.
GE is also talking up Veo, the company's latest CT dose reduction algorithm, which received FDA clearance in August. Veo uses model-based iterative reconstruction (MBIR), and GE claims the algorithm offers a 50% improvement in spatial resolution. MBIR is currently available only on the company's flagship Discovery CT750 HD scanner, while the firm's adaptive statistical iterative reconstruction (ASIR) algorithm is now being migrated to 16-channel scanners in the company's product line.
Other CT highlights include Optima CT660, a compact CT scanner that was cleared by the FDA in August; the company's Dexus workflow and connectivity software, which makes advanced visualization tools available on PACS workstations; and Freedom Edition, a new cardiac scanning package for the Discovery CT750 HD system. Freedom Edition is being shown as a work-in-progress and will be offered as an upgrade.
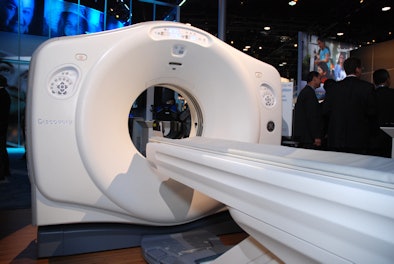 |
| Freedom Edition is a new cardiac scanning package for the Discovery CT750 HD system. |
Healthcare IT
GE is shining the spotlight on version 2.0 of its Centricity Radiology Mobile Access (RMA) platform, which recently received FDA 510(k) clearance for diagnostic use on Apple's iPad and iPhone devices.
RMA 2.0 is cleared for primary diagnosis of CT/MR images when a PACS workstation is not available. The new release features similar functionality as version 1.1 and provides access to images and reports. RMA employs server-side image rendering and does not download personal data to the device, GE said.
GE is also discussing work-in-progress collaboration and electronic medical record (EMR) integration capabilities for RMA.
In addition, the company is emphasizing AW Server, which allows Centricity PACS users to access 3D clinical applications previously available only on GE's Advantage Workstation (AW). GE's Dexus workflow environment, which links imaging devices, clinical applications, and IT, allows users to utilize these 3D tools directly from the Centricity PACS desktop, according to the vendor.
Molecular imaging
In the molecular imaging section of its RSNA 2011 booth, GE is highlighting Discovery PET/CT 710, a system that features 128-slice CT scanning capability, as well as its Q.Suite tools for quantitative PET.
Q.Suite includes Q.Freeze, a work-in-progress application that eliminates motion by combining the quantitative benefits of 4D phase-matched PET/CT imaging into a single static image.
Another Q.Suite element, Q.Static, provides basic motion-correction techniques and automatically isolates data when organs are in a low-motion state, GE said. It then creates a single image series with reduced blurring from organ motion.
Q.AC aims to lower potential variance in attenuation correction measurements, while Q.Check serves as a quality control link between the scanner and the workstation. Q.Core manages both PET and reconstruction processing at faster speeds and the final Q.Suite component, PET VCAR, assists in patient monitoring, according to the vendor.
GE is also discussing development efforts on an integrated PET/MR system as a work-in-progress.
Ultrasound
GE is unveiling Logiq S8, a new portable ultrasound system that can provide coverage of areas such as abdominal, breast, vascular, musculoskeletal, and radiology. It is based on the core of GE's Logiq E9 scanner, but in a smaller package and at an economical price point, GE said.
S8 features include S-Agile Ultrasound, which employs clinically-based models of the body aimed at providing clear images for virtually all body types, according to the vendor. Another feature, Scan Assistant, allows the operator to customize the system to automatically perform frequently used functions. The company is also highlighting the scanner's ergonomic benefits.
 A 3D fetal image acquired with GE's Voluson scanner.
A 3D fetal image acquired with GE's Voluson scanner.
E9 has also been bolstered with a number of upgrades, including color and 4D image quality improvements and an expansion of elastography applications to now include prostate, thyroid, musculoskeletal, liver, and gynecology, GE said. The company has also enhanced the system's volume navigation capabilities and provided enhanced needle-tracking capabilities via its Virtual Needle tracker.
In ultrasound image management developments, GE is presenting version 6.1 of its ViewPoint software. The latest release allows the integration of ultrasound with HIS, RIS, EMR, and PACS, and communication across departments, hospitals, and enterprises, according to the vendor.
Preliminary and final reports can now be created using new "quick reports" templates, and GE's 4D View can be utilized for offline processing, according to the firm.
GE is also showing Nanosonics' Trophon EPR ultrasound transducer disinfection system in its booth.