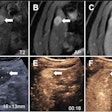
Pharmaceutical developer ImaRx Therapeutics said the first patient has been treated in a new phase I/II clinical trial evaluating the safety and effectiveness of its SonoLysis product for the treatment of peripheral arterial occlusive disease (PAOD).
SonoLysis combines external ultrasound and ImaRx's proprietary nanobubble technology to clear blood clots. The 12-patient multicenter trial will include six patients receiving a 60-minute treatment of SonoLysis (nanobubbles and ultrasound), while the remaining six will receive SonoLysis in conjunction with a small bolus of tPA, a lytic agent administered to dissolve the blood clots that cause PAOD, according to the Tucson, AZ-based firm.
By AuntMinnie.com staff writers
April 12, 2005
Related Reading
ImaRx initiates SonoLysis phase II stroke trial, April 5, 2005
ImaRx raises $7 million, March 10, 2005
ImaRx, Guerbet in MR molecular imaging deal, December 10, 2004
ImaRx completes private placement, names chairman, April 2, 2004
ImaRx inks licensing deal, November 19, 2003
Copyright © 2005 AuntMinnie.com