A daily MR imaging technique can quantify tumor changes in glioblastoma patients undergoing MRI-guided radiation therapy, according to a study published September 30 in the International Journal of Radiation Oncology-Biology-Physics.
The technique -- called MRI-linear accelerator, or MRI-linac -- can serve as an "early warning system" and help clinicians monitor tumor growth during treatment sooner than standard imaging allows, wrote a team led by first author Kaylie Cullison, PhD, and senior author Eric Mellon, MD, PhD, both of the University of Miami's Sylvester Comprehensive Cancer Center in Florida. The group's results were also presented at the recent American Society for Radiation Oncology (ASTRO) meeting.
"Our study shows that these daily scans can serve as an early warning sign for potential tumor growth," Cullison said in a statement released by the university.
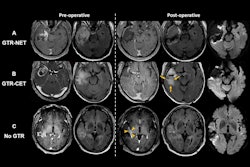
Cullison's group tracked 36 glioblastoma patients over a six-week course of daily radiation and MRI scans using MRI-linac, comparing tumor size data from the daily scans to data from standard-of-care imaging (i.e., a single MRI image with contrast performed one week before radiation treatment and another a month after the radiation course was completed).
The investigators found that for 74% of the 26 patients, MRI-linac imaging results matched those found in the typical contrast MRI performed before and after treatment. For the remaining 26% of patients, MRI-linac imaging predicted tumor growth even though the pre- and post-treatment imaging showed that the tumor shrank.
Even though the daily MRI did not agree with standard MR imaging all the time, "the fact that it did not miss any cases of true tumor growth suggests that it could be used to signal possible tumor growth during radiation therapy," Cullison and colleagues noted.
The technique could also help calibrate radiation treatment, according to the investigators. For tumors shrinking in response to treatment, the radiation field could be narrowed to better target cancerous tissue.
The Cullison and Mellon team plans to perform further research to test MRI-linac's capacity not only to guide treatment decisions for glioblastoma patients during the course of radiation therapy but also in other types of brain cancer, the university said.
The complete study can be found here.