Total tumor volume (TTV) measurements derived from PET/CT imaging can improve survival predictions in patients receiving lutetium-177 (Lu-177) prostate-specific membrane antigen (PSMA)-617, according to a study published March 31 in Radiology.
Researchers at the University of Washington in Seattle first validated previously developed outcome prediction models in a real-world cohort, and then demonstrated that adding TTV measurements improved predictions, noted co-lead authors Lukas Owens and Alireza Ghodsi, MD, and colleagues.
“Incorporating PSMA-PET/CT–derived total tumor volume with existing prediction models for overall survival, progression-free survival, and biochemical response to Lu-177 PSMA-617 improved their discriminatory ability,” the group wrote.
Despite its proven clinical efficacy, response to Lu-177 PSMA-617 (Pluvicto, Novartis) therapy varies significantly among patients with metastatic castration-resistant prostate cancer (mCRPC), the authors explained. This has made accurate outcome predictions a clinical priority, they noted.
Previously developed prediction models incorporating clinical and baseline PSMA-PET parameters have been developed on data from participants in a phase III pre-approval trial, but their utility on data from everyday practice has not been established, the researchers added. To that end, the group sought to validate the models in a cohort of 168 patients treated at their center.
All patients had received at least one cycle of Lu-177 PSMA-617 between June 2022 and April 2024 and PSMA-PET/CT scans within three months before treatment. The median age of patients was 73 years old; 77% had 20 or more metastases and 93% had bone involvement.
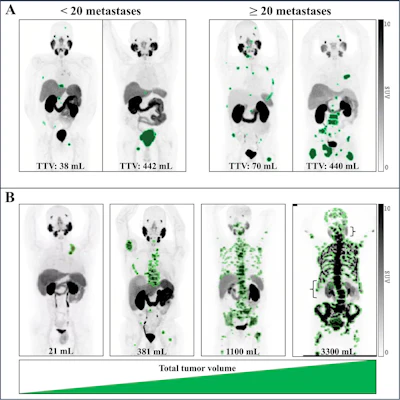 PSMA-PET images. (A) Representative examples illustrate the variability in the total tumor volume (TTV) among patients stratified by the number of metastatic lesions. (B) Case examples demonstrate TTV segmentation across a range of TTV values. Notably, as the tumor volume increased, a visible decrease in radiopharmaceutical uptake by normal organs is observed, particularly in the salivary glands, kidneys, and liver (denoted by brackets), a phenomenon known as the “sink effect.” SUV = standardized uptake value. RSNA
PSMA-PET images. (A) Representative examples illustrate the variability in the total tumor volume (TTV) among patients stratified by the number of metastatic lesions. (B) Case examples demonstrate TTV segmentation across a range of TTV values. Notably, as the tumor volume increased, a visible decrease in radiopharmaceutical uptake by normal organs is observed, particularly in the salivary glands, kidneys, and liver (denoted by brackets), a phenomenon known as the “sink effect.” SUV = standardized uptake value. RSNA
Incorporating TTV increased the C index for OS by 0.04 (P = 0.002), a statistically significant improvement, according to the findings. In addition, higher TTV was independently associated with shorter OS (hazard ratio, 1.35), shorter PSA-PFS (hazard ratio, 1.25), and lower PSA50 rates (odds ratio, 0.70).
"The inclusion of TTV produces prediction models with better discriminatory ability," the authors wrote.
The researchers noted that measurement of TTV requires special software and manual modification, which is time-consuming and may impact reproducibility of the findings. However, deep learning techniques appear to be promising for the task and could help pave the way for the use of PSMA-PET quantitative biomarkers in routine clinical care, they wrote.
“External validation of these refined models incorporating the TTV is warranted,” the group concluded.
In an accompanying editorial, Hebert Vargas, MD, and Sungmin Woo, MD, both of New York University in New York City, lauded the study as a meaningful step in the right direction.
“External validation of nomograms and prediction models on different cohorts than those on which they were developed are key to ensuring their applicability and widespread adoption,” they wrote.
However, to reach the ultimate goal of improving the management of patients with mCRPC, further investigations such as decision curve analysis to determine the net benefit (rather than simply increased calibration of the prediction models), determination of cost and availability of software, and consideration of technical generalizability will be needed, Vargas and Woo concluded.
The full study can be found here.