Dynamic chest radiography (DCR) has huge potential, says a Japanese team leading work on the technique. To show this, the group penned a review that includes more than 20 images illustrating DCR's clinical application in pulmonary vascular diseases.
"Although it has many advantages and clinical utility, most radiologists are unfamiliar with this technique because of its novelty," wrote lead author Yuzo Yamasaki, MD, PhD, of Kyushu University in Fukuoka, in an article published August 26 in the Japanese Journal of Radiology.
DCR is a functional chest imaging technique that requires only three pieces of equipment, according to the authors: a pulsed x-ray generator, a flat panel detector that supports cineradiography, and a dedicated software program for analyzing the images.
"Therefore, DCR can be performed in a general x-ray examination room and has the potential for use as a portable examination system," the researchers wrote.
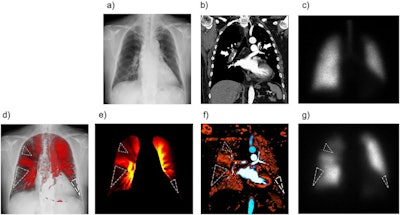 Images of a 50-year-old woman with acute pulmonary embolism. Normal findings are shown in the chest radiograph (a) and anterior planar ventilation scintigraphy (c), whereas the coronal view of the CT pulmonary angiography shows large blood clots in the bilateral pulmonary arteries (b, arrows). Multiple wedge-shaped perfusion defects (dotted triangles) are demonstrated in the bilateral lungs in perfusion images obtained using the cross-correlation method (d) and reference frame subtraction method (e) of dynamic chest radiography, an iodine map created from computed tomography pulmonary angiography (f), and anterior planar perfusion scintigraphy (g). Image and caption courtesy of Radiology: Cardiothoracic Imaging through CC BY 4.0.
Images of a 50-year-old woman with acute pulmonary embolism. Normal findings are shown in the chest radiograph (a) and anterior planar ventilation scintigraphy (c), whereas the coronal view of the CT pulmonary angiography shows large blood clots in the bilateral pulmonary arteries (b, arrows). Multiple wedge-shaped perfusion defects (dotted triangles) are demonstrated in the bilateral lungs in perfusion images obtained using the cross-correlation method (d) and reference frame subtraction method (e) of dynamic chest radiography, an iodine map created from computed tomography pulmonary angiography (f), and anterior planar perfusion scintigraphy (g). Image and caption courtesy of Radiology: Cardiothoracic Imaging through CC BY 4.0.During patient imaging procedures, synchronized, pulsed x-rays are acquired at 15 frames per second for seven to 15 seconds. Patients may be in standing or supine positions and may be asked to perform different breath holds. The sequential images are then processed and provide views of lungs in motion so that clinicians can assess physiological processes, such as perfusion.
According to Yamasaki and colleagues, the use of DCR has been demonstrated in phantoms, animals, and humans with various pulmonary diseases. In their review, they focused on its clinical applications in the following:
- Acute pulmonary embolism
- Pulmonary hypertension
- Adult congenital heart disease
- Pulmonary arteriovenous malformation
- Pulmonary vein stenosis
- Partial anomalous pulmonary venous return
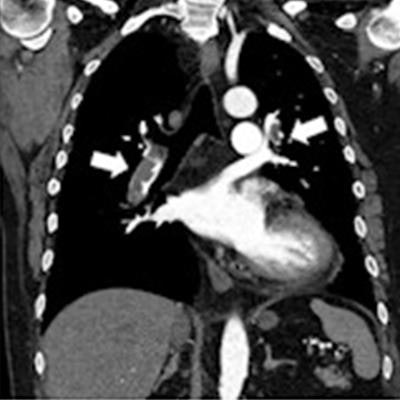
In acute pulmonary embolism -- which is a sudden blockage of the pulmonary arteries by blood clots -- DCR shows triangular or wedge-shaped defects similar to perfusion scintigraphy and the iodine map created from CT pulmonary angiography (CTPA), the authors wrote.
In patients with adult congenital heart disease who develop pulmonary artery stenosis, DCR also demonstrates findings similar to perfusion scintigraphy and invasive pulmonary angiography and facilitates both qualitative and quantitative evaluations of pulmonary circulation, they wrote.
"This convenient and noninvasive method is expected to be an effective alternative approach for assessing lung perfusion in patients with pulmonary artery stenosis," the group explained.
Yet DCR has limitations, the authors noted. For instance, the percentage measurement in the left lower lung may be underestimated in DCR, owing to the shadow of the heart and susceptibility to motion artifacts from heartbeats.
Second, a patient's inability to remain still or hold breath for more than seven seconds as may be required can produce motion artifacts. Finally, while radiation exposure to patients is minimal compared with emissions from CTPA, whether DCR interferes with pacemakers and implantable cardioverter defibrillators remains undetermined, they wrote.
"DCR can evaluate many pulmonary vascular diseases visually and semi-quantitatively and has huge potential in clinical applications," the group concluded.
The full article is available here.