Older people living with HIV show measurably higher brain iron deposits in the motor cortex, and those elevated iron levels are associated with reduced glutamate -- a neurotransmitter essential to normal brain function, according to poster data to be presented May 11 at the ISMRM meeting in Cape Town, South Africa.
Em Triolo, PhD, of Children's Hospital of Philadelphia in Pennsylvania, and colleagues used quantitative susceptibility mapping, an MRI technique sensitive to tissue iron content, alongside MR spectroscopy (MRS) to examine 53 adults: 27 with HIV (mean age, 51) and 26 HIV-negative controls (mean age, 40). The spectroscopy measurements captured concentrations of glutamate, GABA (gamma-aminobutyric acid), and glutathione -- chemicals key to neurotransmission and oxidative stress response.
Among the study participants with HIV, susceptibility values in the motor cortex rose significantly with age, a pattern not seen in their HIV-negative counterparts. The HIV group showed approximately 20% higher susceptibility overall, reflecting greater iron accumulation. When the team tested whether those susceptibility differences tracked with the three measured neurochemicals, only glutamate emerged as significant. The investigators also reported a negative relationship between susceptibility and glutamate concentration across the cohort, meaning participants with higher iron burden tended to have lower glutamate. They did not find any meaningful associations between susceptibility and either GABA or glutathione.
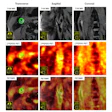
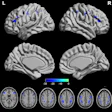
 A) Left (L) and right (R) motor cortex MRS voxel locations (blue), AssemblyNet segmented motor cortex (purple), and overlapping area (green). B) Typical GSH spectrum from edited MRS. C) Typical QSM susceptibility maps.
A) Left (L) and right (R) motor cortex MRS voxel locations (blue), AssemblyNet segmented motor cortex (purple), and overlapping area (green). B) Typical GSH spectrum from edited MRS. C) Typical QSM susceptibility maps.
Em Triolo, PhD, and ISMRM
The team posited that the age-dependent iron increase in people with HIV likely reflects cerebral small vessel disease, a known complication in this population driven by persistent neuroinflammation despite antiretroviral treatment. It noted that iron overload is thought to generate reactive oxygen that damages surrounding tissue, and wrote that the glutamate reduction may represent a downstream consequence of that process, with potential implications for neurotransmission and the cognitive difficulties frequently reported in people aging with HIV.
The work adds to a growing body of evidence that brain iron dysregulation may be an important and measurable mechanism underlying HIV-associated neurocognitive disorders in the modern treatment era, Triolo and colleagues concluded, stressing that these results underscore the need for "future studies with larger sample sizes or broader age ranges."
Check out AuntMinnie.com’s full coverage of ISMRM 2026 on our ShowCast.