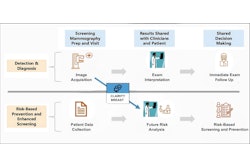
Beth Israel Deaconess Medical Center (BIDMC) in Boston has administered its first clinical cancer risk score using Clairity Breast, a U.S. Food and Drug Administration (FDA)-authorized AI tool for mammogram-based breast cancer risk assessment.
Clairity Breast, which was authorized through the de novo pathway, produces a five-year breast cancer risk score directly from a routine screening mammogram, according to the company. The tool integrates into existing imaging and reporting workflows, allowing providers to order, review, and document results within established systems, Clairity said.
Patients can request a Clairity Breast assessment through BIDMC – Chestnut Hill Square at a routine screening mammogram appointment or through their medical provider, BIDMC said. Results are then reviewed by the provider, with the option to be seen by a breast health specialist at the Breast Care Center at BIDMC if desired.