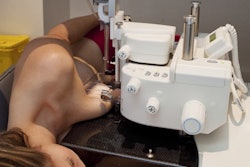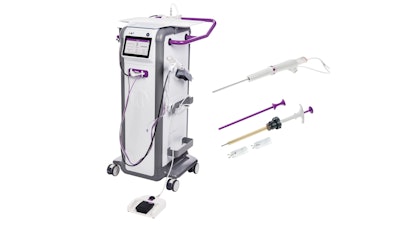
Mammotome has secured clearance from the U.S. Food and Drug Administration (FDA) for two products designed for MR-guided breast biopsy: the Mammotome Prima MR Dual Vacuum-Assisted Breast Biopsy System and the HydroMARK Plus Breast Biopsy Site Marker for MR.
Prima MR is designed to be positioned in the MRI scanner room at the patient's side. It features an 8-gauge needle and touchscreen interface that allows adjustment of aperture size and vacuum strength, according to the company. The system uses 75% less tubing than competing systems, the firm said, enabling larger tissue samples and faster setup and cleanup.
The HydroMARK Plus marker is cleared for exclusive use with the Prima MR system's universal targeting set and incorporates hydrogel technology and titanium shapes that provide visibility under both T1 and T2 MRI sequences, according to Mammotome. The marker is designed to attach to tissue upon deployment and features wings to reduce displacement during surgical excision.
Availability of the products is expected this summer, with international rollout planned for later this year, the company said.