Azurity Pharmaceuticals has launched Ferabright, an iron-based brain MRI contrast agent for adults with known or suspected malignant neoplasms.
The launch follows U.S. Food and Drug Administration (FDA) clearance of the agent in October 2025.
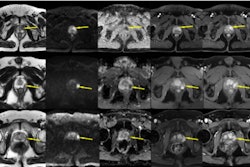
Ferabright is an iron-based contrast agent indicated for brain MRI exams that consists of a superparamagnetic iron oxide nanoparticle with a plasma elimination half-life of about 21 hours. Brain tumor enhancement is best visualized in delayed-phase MRI when contrast marks blood-brain barrier breakdown and macrophage uptake, Azurity said. In a clinical trial, Ferabright-enhanced MR imaging significantly improved visualization of primary and secondary brain neoplasms compared with precontrast imaging.
Ferabright is processed through the body's natural iron metabolism pathways and is available as a contrast option for patients with renal insufficiency or gadolinium contraindications, the company said. The agent carries a boxed warning for anaphylaxis and other serious hypersensitivity reactions, including fatal cases. Administration requires intravenous infusion over at least 15 minutes with monitoring for at least 30 minutes following infusion.
Azurity said Ferabright will be supplied in single-dose vials of 300 mg elemental iron per 10 mL and 510 mg elemental iron per 17 mL.