Healthcare AI startup Ezra has secured 510(k) clearance by the U.S. Food and Drug Administration (FDA) for its Ezra Flash software.
The software will help with the company's goal of launching a 30-minute, full-body MRI scan by helping improve the quality of MR images.
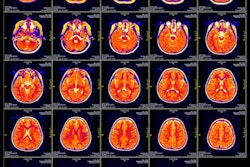
The FDA clearance is for the software's use in brain imaging. Ezra trained Flash AI with a proprietary longitudinal MRI dataset and from there, developed a multi-step validation process.
Ezra's full-body MRI is available in New York, Los Angeles, San Francisco, Miami, and Las Vegas. The company added that it will roll out its services to more cities in the second half of 2023.