Clarity Pharmaceuticals has reported results from the Co-PSMA investigator-initiated trial comparing its copper-64 (Cu-64) SAR-bisPSMA PET/CT agent to standard gallium-68 (Ga-68) prostate-specific membrane antigen (PSMA)-11 PET/CT in 50 patients with prostate cancer.
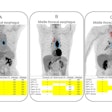
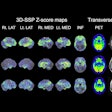
Patients had relapsed disease following radical prostatectomy with low prostate-specific antigen (PSA) levels of 0.2 to 0.75 ng/mL, the company said in a release. Imaging with Cu-64 SAR-bisPSMA identified 63 total lesions compared with 24 for Ga-68 PSMA-11. The true positive rate was 71% for Cu-64 SAR-bisPSMA versus 29% for Ga-68 PSMA-11 and imaging with the copper agent resulted in a management change in 44% of trial participants, the firm said.
The results were presented March 16 at the European Association of Urology Congress 2026 in London and have been accepted for publication in European Urology, Clarity noted.