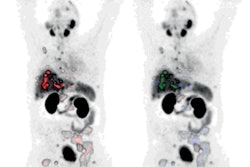
Patients with certain genetic mutations have inferior overall survival after treatment with lutetium-177 (Lu-177) prostate-specific membrane antigen 617 (PSMA-617), researchers have reported.
Specifically, among 72 patients with advanced prostate cancer who had at least one cycle of Lu-177 PSMA-617, those with mutations in key tumor suppressor genes had inferior survival, noted lead author Abigail Pepin, MD, of the University of Pennsylvania in Philadelphia, and colleagues.
“TP53, PTEN, and RB1 mutations were linked to inferior overall survival in Lu-177 PSMA-treated patients and may serve as prognostic biomarkers,” the group wrote. The study was published on February 19 in the Journal of Nuclear Medicine.
Lu-177 PSMA-617 (Pluvicto, Novartis) was approved in 2022 based on the results of the VISION trial, which showed the drug can improve overall survival and radiologic progression-free survival (PFS) in men with metastatic castration-resistant prostate cancer (mCRPC). Yet patients in the trial and those currently being treated have variable responses to the treatment, and thus there is a need for further prognostic and predictive markers, including clinicopathologic and genetic factors, the authors wrote.
To that end, the group characterized oncologic outcomes of patients with mCRPC and genetic aberrations after treatment. Pepin and colleagues analyzed data from 72 patients at their center who underwent germline or somatic genetic testing and received at least one cycle of treatment. Patients were heavily pretreated, with nearly 92% having received taxane-based chemotherapy and 97% having received prior androgen receptor pathway inhibitors, the researchers noted. Germline testing, somatic testing, or both occurred in 87.5% (n = 63), 75% (n = 54), and 59.7% (n = 43) of patients, respectively.
According to the results, while patients with PTEN, TP53, and RB1 alterations did not demonstrate inferior progression-free survival relative to patients without pathogenic mutations (hazard ratio [HR], 2.106), they did experience significantly inferior overall survival outcomes (HR, 3.564).
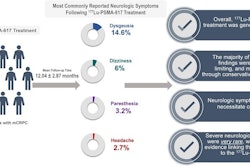
In addition, patients with PTEN, TP53, and RB1 alterations received significantly fewer cycles of Lu-177 PSMA-617 compared with those without the mutations (p = 0.018). In the TP53-mutated group, nearly 80% of patients discontinued therapy early due to disease progression, death, or transition to hospice care.
The researchers noted that patients with TP53, PTEN, and RB1 mutations -- so-called tumor suppressor genes (TSGs) because they inhibit abnormal cell growth -- often harbor significantly higher rates of aggressive cancer pathology. TSG mutations have also been linked to treatment resistance in advanced prostate cancer.
Ultimately, the results of this study warrant further investigation of therapies used for mCRPC patients with mutational burdens, the researchers wrote.
“Prospective validation and larger multicenter studies are required to establish the prognostic and predictive value of germline and somatic aberrations in patients treated with Lu-177 PSMA-617,” the group concluded.
The full study is available here.