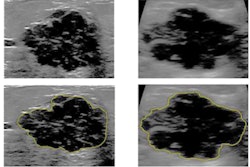
Artificial intelligence (AI) can predict risk for malignant breast lesions just by assessing normal-tissue ultrasound images, suggests research presented at the Society for Imaging Informatics in Medicine (SIIM) annual meeting in Kissimmee, FL.
Researchers led by Dr. Lev Barinov from Memorial Sloan Kettering Cancer Center in New York found that AI-based analysis of normal background parenchymal breast tissue on breast ultrasound could discriminate between the malignancy status of a lesion without having to observe it directly. In testing, the software outperformed the popular Tyrer-Cuzick risk-assessment model.
"These findings clearly demonstrate that AI holds the promise to make clinically meaningful delineations between high- and low-risk background breast tissue on breast ultrasound examination images," Barinov told AuntMinnie.com.
Although previous studies showcase AI's performance in decision support for breast cancer prediction and treatment, they mainly focus on characterizing and classifying soft-tissue lesions and not background parenchymal tissue. However, mammography research also shows there is "significant information" in such background tissue that could help radiologists stratify patient risk and perform lesion-centric risk evaluations, the researchers wrote.
Barinov and colleagues wanted to show that evaluating normal tissue during diagnostic and screening ultrasound exams can assess a patient's risk of having or developing malignant breast lesions. The team looked at 687 cases, of which 197 had examples of malignant lesions. Also out of the total, 39 lesions were deemed to be high risk and 440 were found to be benign.
The AI model (Koios DS Breast, Koios Medical) utilizes a pretrained implementation of ResNet-18 and a combination of features for the network to pool instances of high-risk imaging features. The model also makes use of replication to match ultrasound data with the network's required input dimensionality.
The study authors found that the software achieved an area under the curve (AUC) of 0.66. They wrote that this was consistent with findings from other risk-stratification studies that used mammography, including a 2019 study that employed the current version of the Tyrer-Cuzick model (AUC = 0.61).
"We were pleased to find that the performance of our model exceeds the typical benchmarks of the current version of Tyrer-Cuzick, suggesting it may capture information beyond genetics and estrogen exposure risk factors or that it may more effectively capture associated risk factors," Barinov said.
Barinov also told AuntMinnie.com, "Utilizing machine-learning models, radiologists may eventually have the ability to create significantly more precise breast cancer risk stratifications allowing efficient and appropriate resource allocations to patients with the greatest need."
The team's future goals include follow-up validation research and development for the model, which will involve comparative risk stratification metrics.
"In addition to validating our model, it will also allow us to test the independence of each of the approaches as well as possible fusion mechanisms," Barinov added. "We are all extremely excited about the future prospects for further improving diagnostic accuracy and elevating the quality and cost-effectiveness of care delivery."