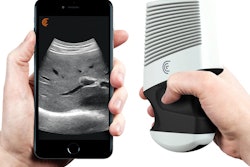
NEW YORK CITY - Ultrasound technology start-up Clarius Mobile Health unveiled a wireless, handheld ultrasound scanner that works with mobile devices at this week's American Institute of Ultrasound in Medicine (AIUM) meeting.
The self-contained handheld scanner communicates wirelessly using a mobile app that is available for both iOS and Android. It is primarily designed for point-of-care applications such as quick exams and for guiding procedures such as nerve blocks and targeted injections, according to the vendor.
 Clarius shows its handheld ultrasound scanner at AIUM 2016.
Clarius shows its handheld ultrasound scanner at AIUM 2016.Linear- and curved-array versions will be available. Clarius intends to file for U.S. Food and Drug Administration (FDA) clearance next month, and the company also plans to submit the scanner for regulatory clearance in Europe and Canada. Clarius declined to provide pricing information at this time.
After obtaining clearance, Clarius intends to offer the scanner via online sales and also through affiliated partners such as manufacturer representatives.