Fluidx Medical has completed enrollment in a clinical trial of its GPX embolic device, and it is highlighting results from a multicenter trial.
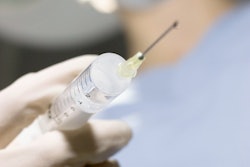
GPX is used to treat primary and metastatic tumors, renal adenoma tumors, and other arterial and venous applications. The company said the device is packaged ready-to-use in a syringe, can be prepared tableside by a clinician in about 30 seconds, and can be delivered through standard microcatheters. The technology is a low viscosity, aqueous-based solution in a syringe that solidifies into an embolus upon delivery without polymerization or dimethyl-sulfoxide precipitation.
Fluidx said GPX has promising applications for tumor embolization. The company has presented interim results at recent annual congresses, including Global Embolization Symposium & Technologies, Society of Interventional Radiology, and Leipzig Interventional Course.
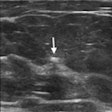
While Fluidx did not disclose exact data from the trial in its announcement, company leaders said GPX showed "significant potential" to advance liquid embolics and demonstrated "excellent" radiopacity.