
Minnies winners, page 3
Scientific Paper of the Year
Minnies 2020 winner: Artificial intelligence versus clinicians: systematic review of design, reporting standards, and claims of deep learning studies. Nagendran M et al, BMJ; published online March 25, 2020. Learn more about this paper.
 Dr. Myura Nagendran.
Dr. Myura Nagendran.It's a frightening notion that artificial intelligence (AI) research might be endangering patients, but that was exactly the main conclusion drawn by the authors of the Scientific Paper of the Year. They found that many studies claiming AI algorithms are superior to human experts for interpreting medical images tend to be of low quality and based on exaggerated claims.
"In many ways, the AI revolution is similar to the genetics revolution of the late 1990s and early 2000s: Misleading information can warp people's expectations and add to the hype among the public and press for accelerated implementation," first author Dr. Myura Nagendran, clinical PhD fellow and intensive care physician at Imperial College London, U.K., told AuntMinnie.com.
Nagendran noted that media headlines such as "Google says its AI can spot lung cancer a year before doctors" and "AI is better at diagnosing skin cancer than your doctor, study finds" were helping create this climate.
"We aimed to suggest how we can move forward in a way that encourages innovation while avoiding hype, diminishing research waste, and protecting patients," he explained.
The idea for the BMJ paper came out of an informal chat over coffee in late 2018. Nagendran and his longstanding friend and colleague Dr. Mahiben Maruthappu decided they wanted to find out how strong the evidence in support of AI really was. Maruthappu is CEO and co-founder of Cera, a home healthcare company and social care provider in the U.K., as well as senior author for the paper.
The duo were convinced that other authors on their paper should comprise experts from a range of different disciplines, so they put together a multidisciplinary team that included the U.S. cardiologist and physician-scientist Dr. Eric Topol and U.K. radiologist Dr. Hugh Harvey, who co-moderated the Artificial Intelligence Exhibition at the 2019 European Congress of Radiology.
The 10 authors then analyzed a group of papers on AI and deep learning that compare diagnostic algorithm performance with human clinicians, evaluating the methods and quality of reporting and transparency of the studies. They submitted the paper to BMJ in October 2019, opting for a general medical journal with a broad readership.
"It can sometimes be a struggle for journal editors to find two or three reviewers for a paper, so it came as a surprise to us that our article had been sent to no less than seven reviewers! It's safe to assume the topic created a lot of interest, and this did mean quite a lot of work for us when it came to making revisions," Nagendran recalled, adding that the paper was finally accepted on 11 February 2020.
The publication date came during the same week as the start of the U.K.'s national lockdown due to the COVID-19 pandemic. Therefore, the response was steady rather than instant and spectacular, he said. Most feedback was positive, although some commentators felt the authors might have been a little too harsh on the innovators.
As of August 2020, Nagendran is now working on his doctoral research, which will explore the interface between AI and clinicians, as well as how to leverage explainable AI for maximal benefit.
Runner-up: Radiology department preparedness for COVID-19: Radiology scientific expert review panel, Mossa-Basha M et al, Radiology, March 16, 2020. Learn more about this paper.
Best New Radiology Device
Minnies 2020 winner: Somatom On.site portable head CT scanner, Siemens Healthineers
Once thought of as a niche application, portable CT is drawing attention as a flexible technology that has a number of advantages over traditional stationary scanners. That interest is reflected in the selection of Somatom On.site by the Minnies expert panel as the Best New Radiology Device for 2020.
Siemens introduced On.site at the 2019 RSNA meeting, where the scanner immediately turned heads. The unit sits on a portable motorized trolley that can be easily moved with a handle, and images can be sent to PACS wirelessly. The 32-slice scanner has a 0.7-mm detector width, providing 2.4 cm of coverage per gantry rotation. On.site received U.S. Food and Drug Administration clearance in July 2020.
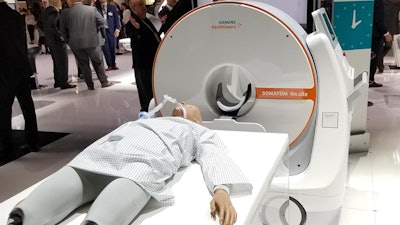 Somatom On.site portable head CT scanner. Image courtesy of Siemens Healthineers.
Somatom On.site portable head CT scanner. Image courtesy of Siemens Healthineers.On.site represents the integration of multiple Siemens technologies into a single system, according to Doug Ryan, vice president of the CT business line for Siemens Healthineers North America. The trolley is based on the company's mobile x-ray systems, while On.site uses the same type of Stellar detectors found on the company's traditional CT scanners.
Siemens developed the system to improve on existing portable CT designs, according to Ryan. While there are some 500-600 portable CT scanners installed globally by other manufacturers, Ryan said that Siemens customers were asking the company to develop its own design that addressed the following three major issues: image quality comparable to a traditional CT scanner, better mobility, and improved radiation safety.
The company has designed the scanner for neurological intensive care units (ICUs), where transporting patients to other hospital departments can have serious clinical consequences: Studies have found that 70% of patients transported outside of the neuro ICU have negative effects, while there is a 13% mortality rate for patients who are moved outside the department.
In addition to clinical consequences, patient transport can also have workflow impacts, requiring anesthesiology, nursing, and ward staff assigned to accompany patients. And neuro patients often require the CT suite to be booked for twice as long as other patients due to their special requirements.
Finally, the COVID-19 pandemic has placed a renewed emphasis on portable imaging, as with mobile systems imaging departments don't have to shut down CT scanning rooms for cleaning and disinfection after a COVID-19 patient is scanned.
"We didn't design this with COVID in mind, but this has a big benefit in this era of COVID," Ryan told AuntMinnie.com.
On.site has just completed a clinical test at Lahey Hospital and Medical Center in Burlington, MA, where clinicians were particularly impressed with the radiation shielding on the system, Ryan said. While Siemens will target neuro ICUs in the first two years of the system's rollout, the company is not ruling out installing the systems in ambulances as part of a mobile stroke service, if a cost-benefit analysis of such installations pans out.
"There is a real need out there, and this creates a brand-new market segment," he said.
Runner-up: Aquilion One / Prism edition CT scanner, Canon Medical Systems
Best New Radiology Software
Minnies 2020 winner: InferRead Lung CT.AI software, Infervision
Chinese artificial intelligence (AI) software developer Infervision was awarded the Best New Radiology Software award for InferRead Lung CT.AI, an AI-based software application designed to help radiologists detect pulmonary nodules on chest CT scans.
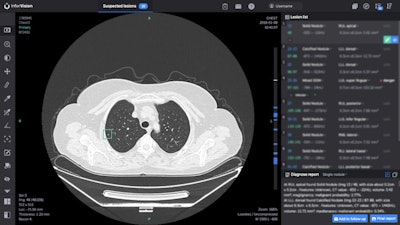 InferRead Lung CT.AI is designed to aid radiologists in detecting pulmonary nodules on chest CT. Image courtesy of Infervision.
InferRead Lung CT.AI is designed to aid radiologists in detecting pulmonary nodules on chest CT. Image courtesy of Infervision.Infervision developed Lung CT.AI after interviewing radiologists and finding out that looking for lung nodules was among their most time-consuming tasks, said Yufeng Deng, PhD, director of Infervision North America.
"This is a time-consuming and repetitive task and it's really a perfect use case for AI," Deng said.
In addition to detecting lung nodules on chest CT scans, Lung CT.AI can show the image slice where the nodule was found, as well as the nodule's location on that slice. It can also provide nodule measurements such as volume and long or short diameter, Deng said.
The deep-learning algorithm was trained initially using data provided primarily from collaborating hospitals in China. As the company began to bring the software to other countries, however, comparison tests showed that the software still performed quite well on datasets from other parts of the world, Deng said.
"We did do some fine tuning [using] public datasets, some from the U.S. and some from other areas," Deng said. "But it was a pleasant surprise that we didn't have to do much work to bring the product from China to other parts of the world."
Deng said that the algorithm has achieved over 95% case-based sensitivity in testing, and it could reduce the number of missed nodules by up to 35% and decrease exam reading time by up to 30%. Lung CT.AI is designed to be used concurrently by radiologists during interpretation.
Initially available for clinical evaluation in China in early 2018, Lung CT.AI received U.S. Food and Drug Administration (FDA) clearance in July and has also received the CE Mark in Europe. In the U.S., Lung CT.AI would be particularly suited for lung cancer screening programs, according to Deng.
Currently the software has been installed in over 10 countries at over 400 hospitals and imaging centers, so far mostly in China. Lung CT.AI is being used on an average of 54,000 exams per day and on over 25 million studies overall, Deng said.
Infervision is also working to add computer-aided diagnosis features such as a nodule malignancy score to Lung CT.AI, he added.
"We want to extend its capability from screening to diagnosis, [and] make it a holistic tool for a lung screening program," Deng said.
Runner-up: Clara COVID-19 AI Classification Model, Nvidia and the U.S. National Institutes of Health
Best New Radiology Vendor
Minnies 2020 winner: RapidAI
RapidAI is an unconventional winner of the award for Best New Radiology Vendor, emerging in February after a rebranding from its previous iteration as iSchemaView, known for its Rapid stroke imaging software.
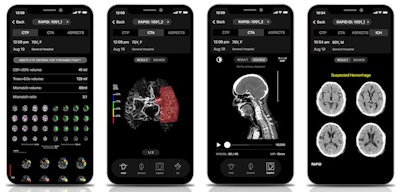 RapidAI's Rapid cerebrovascular imaging AI platform includes a range of stroke imaging and assessment applications. Images courtesy of RapidAI.
RapidAI's Rapid cerebrovascular imaging AI platform includes a range of stroke imaging and assessment applications. Images courtesy of RapidAI.iSchemaView was co-founded in 2008 by neurologist Dr. Greg Albers and radiologist Roland Bammer, PhD, to commercialize the initial Rapid stroke imaging software technology they had developed at Stanford University. The name change to RapidAI reflected the evolution of the company from its ischemic stroke imaging roots to a broader focus on cerebrovascular imaging, as well as its emphasis on artificial intelligence (AI) technology, said president and CEO Don Listwin.
The Rapid imaging platform has expanded significantly over the last few years to include modules such as Rapid CTA and Rapid ICH. In addition, a mobile app version of the Rapid software changed Stanford's stroke practice, said Albers, who is still director and co-founder of the Stanford Stroke Center in addition to serving as co-founder and scientific lead at RapidAI.
"We have about 15 different hospitals that will call us and ask us about transferring patients, and in the past, none of these had any way to get the images to us and they weren't doing more-advanced imaging," he said. "So now, about two-thirds of these referring hospitals have Rapid and then not only can we see their images in real-time on the phone and learn about the patient from the app, but we also get the advanced imaging [analysis] that tells us that there's salvageable tissue or not, which makes it so much easier to decide whether to transfer the patient."
Multiple new Rapid offerings have been launched in 2020. In August, RapidAI garnered U.S. Food and Drug Administration (FDA) clearance for Rapid LVO, its software application for detecting large-vessel occlusions (LVO). Notably, Rapid LVO has also qualified for the U.S. Centers for Medicare and Medicaid Services (CMS) new-technology add-on payment (NTAP) for use of the software in triage and notification of stroke.
In addition, the firm received FDA clearance in July for Rapid ASPECTS computer-aided diagnosis software, which is designed to automatically identify the Alberta Stroke Program Early CT Scoring (ASPECTS) regions of the brain on noncontrast CT and then generate an ASPECTS score to indicate early signs of brain infarction.
Earlier this year, RapidAI unveiled RapidAI Insights, a data analytics platform designed to help users gain insight, standardize their stroke care processes, and optimize operational efficiencies, according to the firm. What's more, the company purchased cerebral aneurysm management software developer EndoVantage and raised $25 million in funding to support further development of its AI software and other potential acquisitions.
To date, Rapid's cerebrovascular imaging platform has been utilized to analyze well over 1 million imaging studies from more than 1,600 hospitals, according to the company.
Looking ahead, Albers said he's excited about the benefits of applying the multiple RapidAI software modules simultaneously, enabling clinicians to gain a lot of information about a patient from a single imaging session. AI can help physicians not miss important findings, such as a subtle hemorrhage that could be overlooked in a patient being considered for a clot-dissolving drug, or an incidental asymptomatic aneurysm that might not be noticed in a stroke patient, he said.
"So it's really the combination of the physician and the software together substantially raising the bar for the level of care that we can now offer," Albers said.
Runner-up: Hyperfine Research
Best Educational Mobile App
Minnies 2020 winner: CTisus iQuiz, Dr. Elliot Fishman (iOS)
 CTisus iQuiz. Image courtesy of Dr. Elliot Fishman
CTisus iQuiz. Image courtesy of Dr. Elliot FishmanDr. Elliot Fishman of Johns Hopkins University has dominated the Best Educational Mobile App award, now landing four Minnies over the eight years since the category was added to AuntMinnie.com's annual competition. This year, CTisus iQuiz joins CTisus Critical Diagnostic Measurements in CT (2016) and CTisus iPearls (2017 and 2019) as winners in Fishman's CTisus family of mobile apps.
CTisus iQuiz, which has been available for a decade, utilizes the quiz -- a classic format for radiology education -- to help users learn about CT. Over 1,000 cases are available on CTisus and each includes the question and correct answer, as well as an audio discussion by Fishman and up to 10 factoids for further reading on the subject.
"So it's really like self-paced learning to images," he said.
The app is organized into 16 different categories. Users can navigate the app either by looking at the most recent month or by going into a category, such as the pancreas, kidney, or liver. For example, there are 160 cases in the pancreas category.
In addition, CTisus iQuiz keeps track of users' progress, recording previous answers and enabling them to return to where they left off the next time they use the app, said Fishman, who also acknowledged Sara Ramin Pour and Lilly Kauffman of Johns Hopkins for their contributions to the project.
As of mid-October, CTisus iQuiz has been downloaded nearly 47,000 times. CTisus iQuiz, like all CTisus apps, is free to download and use. It's updated twice a year, Fishman said.
In the future, Fishman plans to fill out some sections of the app that may not have as many cases as others. He said he's also exploring options for incorporating a little more interactivity, adding different searching functionality that would enable keyword searches for specific types of cases, such as gunshot cases or pediatric cases involving the pancreas.
"We try to think about what people would like," Fishman said.
Runner-up: Radiology Assistant 2.0, BestApps (iOS)
Best Radiology Image
Minnies 2020 winner: 3D-segmented lung image of a COVID-19 patient, Louisiana State University
The origins of this year's winner of the Best Radiology Image category came during a medical imaging scan of an alligator.
That's when radiologist Dr. Bradley Spieler first began working with Emma Schachner, PhD, an assistant professor of cell biology and anatomy, both of the School of Medicine at Louisiana State University (LSU) Health Sciences Center. An evolutionary biologist, Schachner was performing research on reptile lung anatomy and had reached out to Spieler, currently vice chair of radiology research, to see if he could connect her with a private practice that would perform the scans.
Schachner has long specialized in animal anatomy and runs the Schachner Lab at LSU Health Sciences Center, which investigates the origin and evolution of novel morphological innovations in vertebrates. In particular, the lab uses 3D modeling and other techniques to demonstrate how evolutionary processes can affect anatomy.
Fast forward to 2020. The COVID-19 pandemic had begun to hit U.S. shores, and patients were arriving at hospitals with severe lung damage. Both Spieler and Schachner believed that existing tools for imaging the lungs were inadequate to truly visualize fine details of the damage the disease was causing, and they decided to apply to clinical cases the 3D-modeling techniques Schachner had developed in her work.
"We wanted to quantify volumetrically the extent of pneumonia that is normal and what is [acute respiratory distress syndrome], how that affects patient outcomes," Spieler said.
Some adaptations had to be made, however. Schachner can spend weeks working on a single 3D model of an avian lung -- a process that wouldn't translate well to the clinical environment.
In their award-winning paper, published August 18 in BMJ Case Reports, Spieler, Schachner, and colleagues describe how they created 3D digitally segmented models that demonstrate the extent and distribution of COVID-19 disease. First, thin-section 1-mm contrast-enhanced CT datasets were acquired. Then, these datasets were segmented manually and processed with Avizo 7.1 scientific visualization software (Thermo Fisher Scientific), which Schachner uses for evolutionary anatomy research.
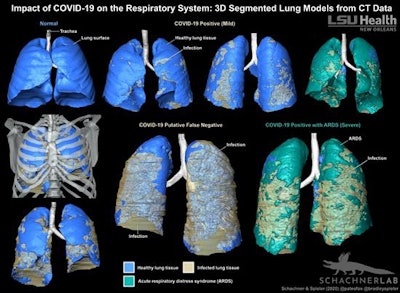 3D-segmented models of lung CT data show the distribution of COVID-19-related infection in the respiratory system. Image courtesy of LSU Health New Orleans.
3D-segmented models of lung CT data show the distribution of COVID-19-related infection in the respiratory system. Image courtesy of LSU Health New Orleans.The researchers acknowledged that their method is more time-consuming than automated volume-rendering techniques, but it produced a "highly accurate and detailed anatomical model where the layers can be pulled apart, volumes quantified, and it can be 3D printed."
Given the time constraints of a busy hospital, does the technique have clinical potential?
Perhaps not for acute care, but Spieler believes it could have uses in areas that aren't so time-sensitive, such as surgical planning. Spieler and Schachner are planning follow-up studies to see if the technique could be used for prognostic applications, such as determining how long patients might stay in the intensive care unit or whether they will need a ventilator.
Runner-up: F-18 FDG PET/MRI image of chronic pain
Previous page | 1 | 2 | 3



















![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)