CHICAGO - New scanner launches at the RSNA 2016 booth of Siemens Healthineers include a CT platform and MRI system, while other highlights range from a robotic interventional radiology system to classical x-ray.
CT
Siemens is making a big splash in CT with the launch of a new product platform of scanners, called Somatom go, that can be controlled using tablet computers. The first scanners in the family are Somatom go.Now, a 32-slice system, and Somatom go.Up, a 64-slice model.
The Somatom go models were developed after an extensive customer feedback process that solicited suggestions on challenges facing Siemens customers. Many of the issues revolved around reimbursement cuts, as well as staff training and education issues and scanner operating costs.
 Somatom go.Now is one of a pair of new tablet-controlled CT scanners.
Somatom go.Now is one of a pair of new tablet-controlled CT scanners.Using a tablet to operate the scanners addresses many of these issues by enabling a mobile workflow in which the technologist is freed from the console and can move around in the scanning room, keeping in close proximity to the patient while controlling scanner functions.
The tablet can control a number of key scanner functions, including patient registration and scanner protocol selection, image reconstruction, and the viewing of reference images for quality assurance.
Both Somatom go.Now and Somatom go.Up employ the company's Stellar detector technology and tin filtering for radiation dose reduction. They also support sinogram affirmed iterative reconstruction (SAFIRE), the company's iterative reconstruction-based dose reduction protocol, and Care Dose4D real-time anatomic exposure control.
Both Somatom go scanners are pending 510(k) clearance from the U.S. Food and Drug Administration (FDA). Siemens expects to begin shipments in the second quarter of 2017.
MRI
For MRI, Siemens is touting Magnetom Sempra, which is targeted to radiological practices, as well as small and medium-sized hospitals and imaging centers. The system is pending 510(k) clearance from the U.S. Food and Drug Administration (FDA). The company hopes to attract customers with amenities such as the scanner's lower consumption of energy and greater uptime through two additional years of coverage from the Healthineers Connect Plan service package.
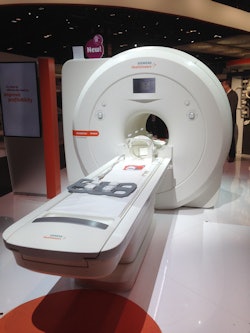 Siemens is demonstrating its new Magnetom Sempra MRI scanner.
Siemens is demonstrating its new Magnetom Sempra MRI scanner.Siemens' DotGo MRI software on Sempra is designed to reduce repeat scans due to operator error and inexperience and the vagaries of diverse patient populations, which can adversely affect exam quality. With the software, clinicians can standardize MRI scans and adjust to the condition of each patient. Sempra will also feature the company's Dot (Day optimizing throughput) engines to automate and streamline workflow when examining the brain, spine, and large joints, which account for approximately 75% of MRI scans. A complete brain scan from patient positioning on the table to exam completion could be completed in approximately 10 minutes, Siemens said.
To help reduce overall operating costs, Sempra includes Eco-Power technology to monitor and control magnet-cooling helium. In addition, Zero Helium boil-off technology is designed to prevent helium evaporation and can avoid refills and downtime.
Siemens also provided a glimpse of its coming Compressed Sensing technology, designed to shorten MR image acquisition times. The first application for Compressed Sensing, which is pending FDA 510(k) approval, will be cardiac cine imaging. Scans are expected to take 16 seconds rather than the conventional four minutes, due largely to a novel algorithm that reduces the amount of data required and the elimination of breath-holds.
Siemens also expects Compressed Sensing Cardiac Cine to benefit patients with cardiac arrhythmias. Adaptive triggering is designed for the entire cardiac cycle, including the late diastolic phase, to be recorded in real-time with a single breath-hold. Compressed Sensing Cardiac Cine also offers enhanced visualization and quantification of left-ventricular function, according to the company.
Molecular imaging
In this area, Siemens is displaying the new version of its xSPECT Quant software, which allows users of the Symbia Intevo SPECT/CT system to perform automated quantification of technetium-99m, as well as iodine-123, lutetium-177, and indium-111. This expansion of quantifiable isotopes advances the use of SPECT quantification from general nuclear medicine and bone studies to indications such as neurological disorders, neuroendocrine tumors, neuroblastoma, and prostate cancer.
xSPECT Quant also uses a 3% National Institute of Standards and Technology (NIST) traceable precision source to standardize uptake values for disease detection and therapy response across imaging centers, cameras, and dose calibrators. The quantification capabilities can also facilitate theranostics, an approach that combines diagnostic and therapeutic efforts.
X-ray
In x-ray, Multix Fusion Max is a new midrange ceiling-suspended x-ray room. The system is designed for a full range of clinical applications, including general x-ray, trauma and emergency, chest screening, on-table examinations, and high-throughput general x-ray indications, and it is targeted at small and medium-sized hospitals.
Multix Fusion Max users can swap flat-panel detectors from the unit between other systems in the Max family, a feature designed to improve system uptime while supporting the availability of backup units. The company's MaxCharge feature prevents battery-related interruptions by charging the unit's Max wi-D detector whenever it's inside the table or bucky wall stand, as well as when it's stored inside its mobile detector holder.
Users can also image patients from head to toe while the patient is standing, sitting, or on a mobile table, further enhancing the system's flexibility. In addition, when the height of the table or wall stand is adjusted, the x-ray tube of Multix Fusion Max tracks and synchronizes automatically to preserve the correct source-to-image distance. Multix Fusion Max has U.S. FDA clearance.
For the Multitom Rax twin robotic x-ray room, first launched at the RSNA 2015 meeting, Siemens is touting the achievement of an installed base in the high double digits. A 3D functionality will be available on the unit in 2017.
Interventional x-ray
Artis pheno is a new angiography system that features the company's robotic technology and is designed to meet the growing need for hybrid operating rooms in interventional radiology, minimally invasive surgery, and interventional cardiology. The system sports a zen40HDR flat-panel 30 x 40-inch digital detector and the company's Gigalix x-ray tube, as well as a new 2K imaging chain that offers 2D resolution four times higher than previous Siemens systems.
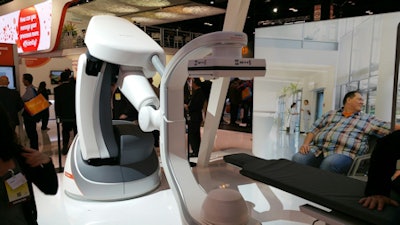 Artis pheno is a new robotic-controlled interventional x-ray system.
Artis pheno is a new robotic-controlled interventional x-ray system.A feature called StructureScout enables users to adapt and optimize imaging parameters to suit the region of interest, offering the potential for reduced x-ray dose compared to previous Siemens products. Optional software packages are also available to give users the ability to treat patients with complex conditions or multiple comorbidities.
Siemens also put a major emphasis on infection control when designing Artis pheno; the system has large, sealed surfaces with fewer spaces, making it easy to clean and disinfect.
Artis pheno is also faster than the preceding Siemens angiography system, Artis zeego, with a C-arm that is 5 inches wider and a free inner diameter of 37.6 inches. This gives users more space for handling adipose patients and enables instruments to be used for longer periods. Meanwhile, the system's table supports patients up to 617 lb.
Artis pheno is also faster; a DynaCT brain exam that used to take 20 seconds now can be performed in only six seconds, due to the unit's speed of 90 frames per second. The system also has an integrated tableside ultrasound scanner, thanks to Siemens' wireless ultrasound transducer technology.
Siemens has applied for FDA clearance for Artis pheno, and the system is being shown as a work-in-progress at RSNA 2016.
Also in angiography, Siemens has added new features to its family of angiography systems, Artis zee, Artis Q, and Artis Q.zen. Syngo EVAR Guidance provides automated detection of vessel walls on CT datasets, and it also supports automatic placement of landmarks for 3D image guidance. It also can suggest the optimal angulation of C-arms for better deployment during interventional procedures. Meanwhile, syngo CTO Guidance automatically segments coronary CT angiography (CCTA) images in addition to providing procedural guidance in cases of challenging chronic total occlusions.
Mammography
In mammography, Siemens' flagship system is Mammomat Inspiration Prime, which is capable of both conventional 2D and digital breast tomosynthesis imaging. Siemens is talking up the low radiation dose on the unit, which the company believes is 30% lower for 2D mammography.
Siemens received FDA clearance in April for the use of Inspiration Prime for screening-only tomosynthesis. It is awaiting word from the FDA on synthetic 2D tomosynthesis, which would obviate the need to perform a 2D acquisition along with 3D volumes for screening.
In addition, the company's enhanced multiple parameter iterative reconstruction (EMPIRE) is a new data reconstruction algorithm for mammography that uses iterative reconstruction rather than filtered back projection.
Ultrasound
In ultrasound, Siemens is showing enhancements to its Acuson S family of scanners, and the company is highlighting a recent study that gave its products high marks for usability. The company believes that liver imaging will become an important new application for ultrasound with the approval of the first ultrasound contrast agent for radiology use, Lumason from Bracco. The company is also highlighting the next generation of its Acuson Freestyle wireless ultrasound transducer.
Imaging informatics
In imaging informatics at RSNA 2016, Siemens is discussing a new version of its syngo.via software that provides better usability and customized fast access to individual user preferences. Syngo.via manages diagnostic findings to make relevant data immediately available to radiologists.
In addition, syngo.via enables the use of cinematic volume-rendering technique (cinematic VRT) with any diagnostic display, giving users photorealistic clinical images generated from any CT or MRI scan. Cinematic VRT images can be used to communicate with referring physicians and patients, as well as for educational and publication purposes.