Breast ultrasound is coming into its own, especially with new attention being paid to dense breast tissue. Breast ultrasound specialists have a range of high-tech tools at their disposal, but sometimes getting back to basics may be the best choice, according to an April 13 talk at the National Conference on Breast Cancer (NCBC), sponsored by the American College of Radiology.
Ultrasound scanners are bundled with all kinds of special features, some of which are not only unnecessary for breast imaging but also may even have a negative effect on image quality, according to a Friday morning presentation at NCBC by Dr. Bruno Fornage of the University of Texas MD Anderson Cancer Center. Fornage outlined which features in breast ultrasound technology are indispensable, which ones are useful, and which ones are flat-out unnecessary.
High-resolution transducers
Advances in ultrasound equipment used for breast imaging include high-frequency and matrix linear transducers that operate at peak frequencies of up to 18 MHz and provide exquisite spatial resolution, according to Fornage. These transducers are now found on most scanners, and they can visualize cancers as small as a few millimeters. But it's also a good idea to have a less fancy transducer on hand, he said.
"It's also important to have a linear-array transducer of a lower frequency, even an abdominal-type curved-array low-frequency probe, so that clinicians can penetrate very large breasts and image lesions that cannot be imaged with the high-frequency probes whose fields of view rarely exceed 4 cm," he said.
Extending field-of-view
Extended field-of-view technology allows operators to "stretch" the standard sonogram to build a static picture with a much wider field of view than that which is available with standard real-time transducers, Fornage told session attendees.
The advantage is the ability to scan through a lesion and the nipple; measuring the distance between the two allows for precise correlation with the location of that lesion on mammograms. Some high-end scanners can steer the beam electronically and "expand" the rectangular field of view of the linear-array transducer into a trapezoidal one.
"This expansion of the width of the field of view in depth may be sufficient to entirely encompass a lesion measuring up to 5 cm," Fornage said.
Powerful Doppler
High-frequency grayscale ultrasound and power Doppler ultrasound (PDUS) are the two tools required to perform an excellent breast ultrasound examination, according to Fornage.
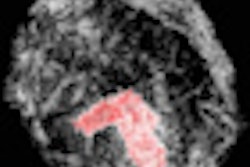
"A simple, yet powerful application of PDUS is differentiating between a rounded, markedly hypoechoic tumor and a [thickened] cyst with low-level internal echoes," he said. "The detection of the smallest vessel within the lesion automatically excludes a cyst -- or any other fluid collection -- and confirms a neoplasm. This very simple yet very useful application of PDUS is still underused."
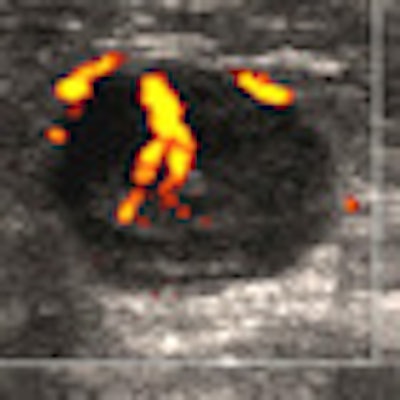
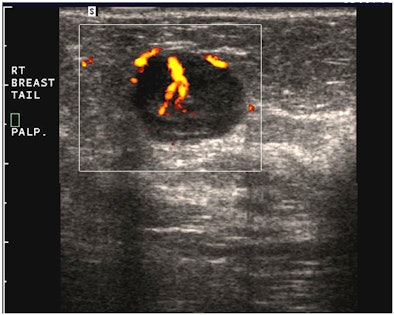
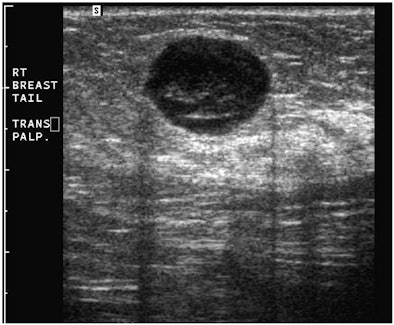 |
| Images are of a circumscribed round pseudocystic mass. Above, grayscale US shows a cystic appearing mass with some low-level internal echoes. Below, PDUS performed with only minimal pressure applied through the probe shows internal vascularity, thereby excluding a fluid-filled lesion and confirming a tissular mass. Fine-needle aspiration readily confirmed a metastasis from a uterine leiomyosarcoma treated six years previously. All images courtesy of Dr. Bruno Fornage. |
 |
Compound scanning
Real-time compound scanning combines the echoes obtained from multiple lines of sight at various angles of ultrasound exposure, compared with the single vertical beam in a standard linear-array transducer, in an attempt to improve the visibility of lesions. But is this technique necessary?
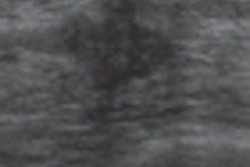
"Due to marketing efforts by some manufacturers, this type of image processing is now a default setting on most scanners sold in this country," Fornage said. "But evidence-based studies proving the superiority of this technique over standard sonography in the detection or characterization of breast lesions are still missing. In fact, the majority of breast ultrasound studies brought in from outside MD Anderson by patients referred to our institution have been performed with real-time spatial compounding and are at times useless because of the blurring affecting them."
Another drawback to this type of image processing is the disappearance of "good" artifacts, such as sound-through transmission, shadowing, and comet-tail artifacts, according to Fornage.
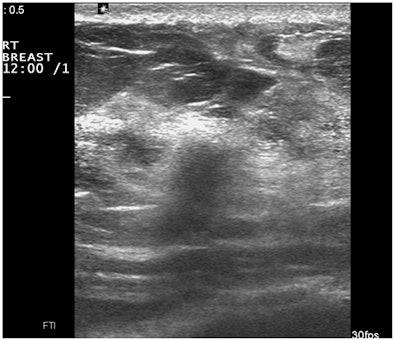 |
| Images show deterioration with the use of spatial compounding. Above, sonogram of a small carcinoma obtained with spatial compounding turned on. Below, sonogram of the same lesion obtained with conventional grayscale US settings without spatial compounding. |
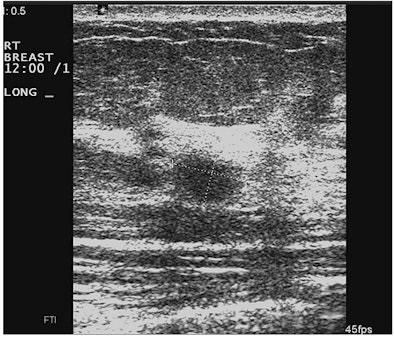 |
Taming tissue harmonics
Tissue harmonic imaging (THI) can increase the spatial resolution and boost the contrast of an ultrasound, and it may improve the delineation of poorly defined lesions. But a major downside of this technology is that the substantial increase in contrast throughout the sonogram may create deceiving areas of shadowing, Fornage said.
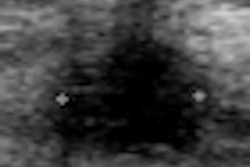
"It's critical to turn this option off after its use so that the next user of the machine will not be tricked by the resulting over-contrasted images," he told session attendees. "THI should be used as an optional tool, not a default setting."
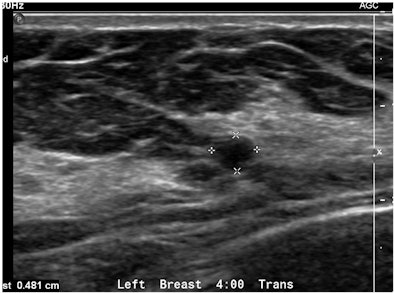 |
| Sonograms from a patient who came to MD Anderson with a BI-RADS 4 US report describing a 0.6-cm solid mass requiring a vacuum-assisted US-guided biopsy. Above, outside study performed with spatial compounding fails to demonstrate the enhancement distal to the minute cyst. Below, examination performed without spatial compounding and with tissue harmonic imaging clearly shows that the mass is a simple cyst. |
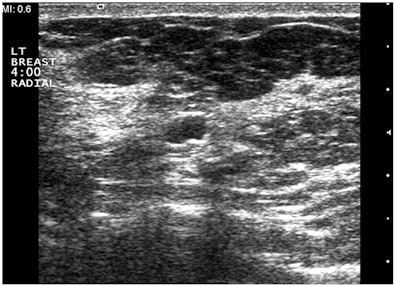 |
3D ultrasound
Three-dimensional ultrasound has been available since the mid-1980s, according to Fornage: The theoretical benefit of the technology is that it permits the reconstruction of sonograms that are otherwise impossible to obtain, especially coronal views. But 3D ultrasound has its limitations, he added.
"The major limitation is the time taken to render the 3D images and, more importantly, to navigate through the multiple additional possible oblique scan planes with the risk of becoming rapidly disoriented," he said. "In fact, the experienced sonographer actually does a very fast 3D ultrasound rendering when he or she scans over a lesion in conventional 2D ultrasound, as the brain quickly integrates the multiple 2D images to create a 3D rendering of the lesion's volume and shape."
Automated breast ultrasound
Automated breast ultrasound (ABUS) is based on a single, very long high-frequency (14 MHz to 16 MHz) linear-array transducer, housed in a pad that is manually applied to the breast, Fornage said. The transducer scans over the breast for a few seconds, acquiring the breast volume dataset and displaying multiplanar images on a workstation.
"[The advantages of this technology] include fast, automatic volume acquisition and reconstruction of coronal views. But limitations in correlation remain, owing to the differences in breast positioning between ultrasound and mammography," he said. "Other limitations include the fact that the device cannot scan the entire breast -- especially if the breast is large -- so that more than one volume acquisition is needed to ensure that the entire breast has been scanned, and that the technology lacks real-time capability, and therefore can't clear an area affected by an artifact."
Contrast-enhanced US
Contrast-enhanced ultrasound (CEUS), especially with the latest-generation ultrasound contrast agents, has proved quite useful in the examination of some internal organs. But for breast masses, the technology does not eliminate the need for a percutaneous needle biopsy -- and it adds another layer of complexity and cost to the workup that is just not warranted, Fornage said.
"CEUS is a research tool that has not made its way into the daily practice of breast imaging," he said.
Elastography: elusive?
Elasticity imaging with ultrasound, or elastography, has become a popular topic in the radiology community, although it has been in the works for more than 20 years with ultrasound and has been available with MRI for many years, Fornage said.
The technology's goal is to provide information related to the stiffness of tissues. But current commercially available scanners are confounded by a lack of intraobserver reliability, so that it's not unusual to produce an opposite result on repeat testing a few seconds later, according to Fornage.
"There are very few evidence-based nonindustry-sponsored studies reporting substantial superiority [of elastography] over standard grayscale ultrasound," he said. "In fact, a sensitivity of 82% in the diagnosis of breast cancer has been reported for elastography, versus 94% for conventional grayscale ultrasound. More disturbing is that even if the technology of elastographyworked flawlessly, the huge overlap in breast pathology between very firm solid benign lesions and less-firm malignancies gives this technology no practical place in the differential diagnosis of solid breast masses."
Back to basics
The truth is that many breast imagers still aren't taking full advantage of all the capabilities of grayscale and power Doppler ultrasound imaging, and are instead limiting their use of ultrasound to merely guiding percutaneous needle biopsies, Fornage concluded.
"Breast imagers should focus on using the time-proven effective basic imaging techniques, be aware of the possible disastrous effects of some oversold image-processing techniques, and refrain from turning to unproven new technologies that will not avoid an 'unnecessary biopsy' but, on the contrary, may very well lead to one," he said.