AuntMinnie.com presents the sixth in a series of columns on the practice of ultrasound from Dr. Jason Birnholz, one of the pioneers of this modality.
Fellow UltraSounder,
Palpation on steroids. Dr. Brian Garra used this phrase a few years ago to describe elastography when I interviewed him for ReachMD as part of the Advances in Medical Imaging series. It's a truly great expression that speaks directly to anyone who has tried to learn the art of physical diagnosis.
 |
| Dr. Jason Birnholz. |
Extreme limitations
The earliest form of elastography to be available in commercial equipment required the user to press the transducer, and then the software would color code and superimpose a few image frames. The concept and initial work was done by Dr. Jonathan Ophir and colleagues (Ultrasonic Imaging, April 1991, Vol. 13:2, pp. 111-134). The idea was to identify what does not move, such as an infiltrated superficial lymph node, which is hard and fixed in place. This technique was really more of an indication of tissue fixation than a map of the elastic features of a tissue field.
This form of elastography has extreme limitations, and it is an absolute credit to the method's proponents that so much great clinical work has been done with it. First, it is uncalibrated, unstandardized, qualitative, and subjective. It is only applicable superficially, and the findings are relative not absolute, meaning that everything depends on the relative values of the target and tissue that surrounds it. It is not useful for diffuse changes in a tissue field, such as identifying or grading cirrhosis.
Most of the work has focused on larger superficial breast tumors where there is a clear physical difference between cancer collections and breast fat that surrounds the abnormal parenchymal field.
I never had much interest in this form of elastography because I never thought it added anything to just watching a lesion continuously during some maneuver. One of the old-time ultrasound imaging tricks was to watch the liver when a patient sniffs forcefully -- is the liver Jell-O or a rock? And, at the softness end of the scale, it is the squish that is important, not the fixation. An example of this is the softening of the cervix that precedes effacement, the earliest risk indicator for premature labor.
Strain imaging
Before our ReachMD interview, Garra had reported with Dr. Jonathan Ophir and others about the potential of strain imaging for differentiating benign and malignant breast tumors (Physics in Medicine and Biology, September 7, 2008, Vol. 53:17, pp. 4809-4823). This form of elastography seeks to adapt the technique for MR elastography in the mid-1990s and is based on the work of Nightingale and colleagues reported in 2001 and reviewed last year (Current Medical Imaging Reviews, November 1, 2011, Vol. 7:4, pp. 328-339).
This method is referred to as acoustic radiation force imaging. The transducer generates a push pulse and minute tissue displacements along the beam axis are determined at the focal point of the push. The strain modulus is then calculated. Harder areas have less displacement and softer regions move more. As this has not yet received U.S. Food and Drug Administration (FDA) clearance, I have no experience with it.
I have been fortunate, however, to be able to use another approach referred to as shear-wave elastography (SWE) in my clinical practice, and I keep appreciating new facets of its use in routine scanning.
Earthquakes and elastography
One way to approach ultrasonic shear-wave elastography is through earthquakes, which is also historically correct because seismology is a foundation discipline for vibrational wave analysis. The mechanical event of the quake produces a shock wave that propagates through the earth. This is the seismologists' P wave for "primary" or "plane." This is a longitudinal wave, exactly like the pulses used for ultrasound imaging. These waves travel by compression and rarefaction of matter along and parallel to the direction of the wave.
After a pretty long interval, seismographs near to the epicenter will record a secondary or S wave. These waves travel a lot slower than P waves and have much lower frequency (longer wavelength); they are transverse waves because they propagate by matter compression and rarefaction at right angles to the direction of the wave. These are also called shear waves or elastic shear waves.
Plane waves may generate shear waves via mode conversion interactions. This is the basis for a lot of nondestructive testing for flaws in manufactured materials, but it has been ignored in clinical ultrasound work because these low energy events do not affect conventional ultrasound image formation.
Real-time shear-wave elastograms
There are two factors that make SWE possible and practical. The first is that certain-shaped, short-time high-frequency component acoustic pulses in the low MHz range generate polarized shear waves in body tissues. The second is that fast plane waves (like those of B-mode imaging) can be used to make a movie of the passage of slower transverse waves. This is done in real-time with the high-speed techniques for 4G ultrasound described in my previous AuntMinnie.com column.
High-speed plane-wave imaging identifies the minute tissue changes as the transverse wave front moves through the region of interest. The velocity of the transverse wave front through tissue is related directly to elasticity. The result is simultaneous display of the B-mode image with a superimposed color-coded map of elasticity wherever the region of interest is placed in the imaging field.
The measure of elasticity is absolute, not relative, so the method can be applied for local disease states such as tumor nodules or atherosclerotic plaque characterization and for diffuse parenchymal changes or even tissue state changes such as resting versus contracted muscle. In the U.S., the method is used semiqualitatively, although quantitative determination of elasticity is inherent in the method and used in that way in Europe and Asia.
SWE derives from the works of Fink, Tanter, Bercoff, and others (Journal of the Acoustical Society of America, December 2007, Vol. 122:6, pp. 3211-3219). Professor Mathias Fink, director of the Langevin Institute of the École Supérieure de Physique et Chimie Industrielles de la Ville de Paris, uses the term "multimodality imaging" for combining unique information from different types of waves samples of tissue into one image. He refers to SWE as a combination that adds a new level of contrast resolution to the superb spatial resolution of B-mode imaging, each contributing something "invisible" with either alone. This is the one-modality equivalent of fusion imaging.
The traditional cyst vs. solid conundrum
Fluid pockets in tissue do not support shear waves. Cysts appear as voids in shear-wave elastograms. An unusual application is to confirm that a fluid structure with a lot of superimposed noise really is a cyst or that a nonclotted blood filled endometrioma is not a solid mass. One of the prices of high-frequency supersensitive ultrasound imaging is that speckle noise increases, and it is devilishly hard to eliminate without losing low-level grayscale details.
Tumors and palpation and the determinant of elasticity
Most people start their elastography experience with tumor applications, particularly the breast or thyroid. The notion is that if a mass looks bad and is hard, it is bad. And if it looks OK and is soft, it may not even need to be biopsied. Generally, we have a greater worry about breast nodules than those of the thyroid, where most are benign, and most of those that are malignant histologically have very slow growth rates.
The difficulty is that not all cancers are hard and, conversely, some soft lesions seem to be hard in elastograms. This is the fallacy of thinking of elastograms as a form of palpation, which channels understanding along lines of hardness or nothing. Put another way, either elastograms are often wrong, or maybe it is something other than hardness that we are mapping.
When I first heard about the phenomenon, I wondered if it had anything to do with local temperature gradients. After all, metabolically active tumors such as triple-negative breast cancers are surrounded by insulating fat and tend to be a few degrees warmer than surrounding tissue.
Professor Fink assured me in the politest way, however, that temperature is not a differential factor for longitudinal and transverse ultrasound waves on theoretical grounds, and he then showed me experimental data demonstrating that elasticity changes require the massive thermal shifts (and protein denaturation) of thermal ablation (Physics in Medicine and Biology, March 21, 2010, Vol. 55:6, pp. 1701-1718). I went to the penalty box recalling that Young of Young's modulus of elasticity was a physician.
Figure 1 is an elegant high-frequency image of a kidney in a newborn infant. I selected this because the grayscale differential between cortex and pyramids is pronounced.
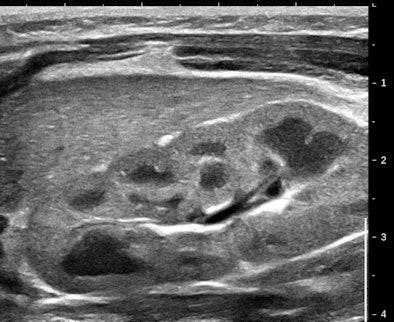 |
| Figure 1. |
All of ultrasound imaging is based on mapping elasticity gradients. The principal determinant of elasticity in tissue is collagen and more precisely the amount, type, and arrangement of collagen fibers. Pyramids and cortex have about the same amount and type of collagen.
The random collagen arrangement of the cortex and the vertical colonnades of the pyramids are a structural factor that accounts for a lot (but not all) of the grayscale and elastic differences. We should expect that processes with fibrosis (or sclerosis) will be harder in elastograms.
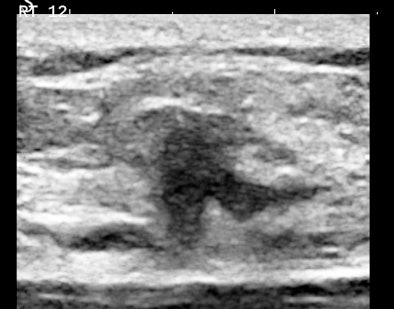
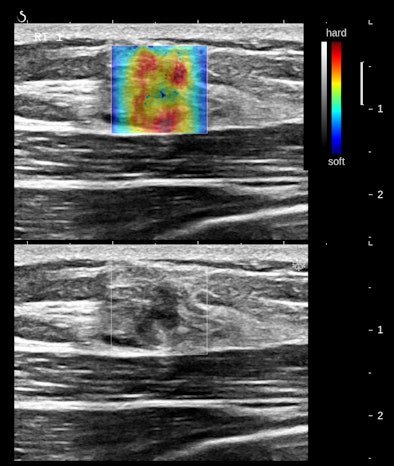
Figures 2A and 2B are cropped B-mode and simultaneous SWE of an 8-mm nodule in an isolated island of parenchyma in the upper outer quadrant of a 43-year-old woman.
 |
 |
| Figures 2A and 2B. |
The B-mode features are very abnormal, at the least BI-RADS 4. The elastogram is particularly interesting because the most abnormal area is the halo surrounding the small, peculiarly shaped nodule. This is the desmoplastic border of hard fibrous tissue that the body is erecting to delay tumor expansion. One might infer from this that this tumor has invasive features that are not apparent in the B-mode image. The pathology was microinvasive ductal carcinoma.
The principal determinants of elasticity that are mapped by shear-wave elastography appear to be cell density, interstitial pressure, and fibrosis. Very vascular nodules contribute both increased collagen from vessel walls and interstitial pressure from circulating blood. The important consideration is that these determinants of elasticity are cellular, below the level of resolution of B-mode imaging. SWE would appear to have the potential to reveal some cellular features of tissues.
Heterogeneous or homogeneous?
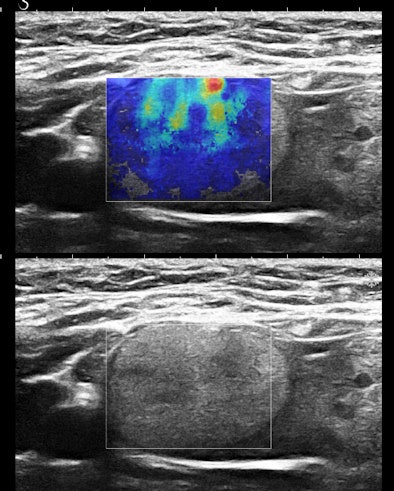
Figures 3A and 3B are the B-mode and simultaneous SWE of a follicular neoplasm of the thyroid. Notice how the elastogram indicates a cell population heterogeneity that is not visible in the original image.
 |
 |
| Figures 3A and 3B. |
Homogeneous and heterogeneous are classic ultrasound image features. Perhaps ultimately, refining this descriptor and even quantifying it will be the most important role of SWE in practice.
For now, let us forget about the mosaicism in some livers that results in geographic patterns of fat deposition in the liver and concentrate on the typical patient with diffuse fatty infiltration. These livers are soft, visually recognized by increased reflectivity without increased attenuation (and they have a lower sound-wave propagation velocity than normal). The really important clinical feature is identifying those with an inflammatory component that are progressing into fibrosis.
A similar issue is in the transition from alcoholic, viral, or chronic active hepatitis into cirrhosis. The main imaging feature of cirrhosis is the change in B-mode architecture of the liver. As you might suppose, SWE is superb at revealing even the onset of periportal fibrosis. This is emerging as one of the most important and reliable applications of SWE in practice and is the subject of scientific papers now in press and in preparation.
I tend to use SWE just like I use Doppler, that is, whenever I think it may help me. Like Doppler, all it takes is depressing a button on the control panel during continuous B-mode imaging. If it helps, it is invaluable, and if it is noncontributory, then all I have expended is an extra few seconds of scanning time. When the determinants of elasticity within tissue are appreciated, it becomes straightforward to relate the image appearances to pathology. Conversely, an appreciation of tissue pathology will also suggest situations in which elastography will be especially informative.
Dr. Jason Birnholz is a graduate of Johns Hopkins School of Medicine and did his diagnostic radiology residency at Massachusetts General Hospital. He was awarded an advanced academic fellowship from the James Picker Foundation and has been a professor of radiology.
The comments and observations expressed herein do not necessarily reflect the opinions of AuntMinnie.com, nor should they be construed as an endorsement or admonishment of any particular vendor, analyst, industry consultant, or consulting group.