PHILADELPHIA - Although approved for radiology applications in Europe, Canada, and Asian countries, ultrasound contrast agents have yet to garner clearance for non-cardiac use in the U.S. However, contrast-enhanced ultrasound (CEUS) can add critically valuable information without adding patient risk in applications such as detecting and characterizing liver and extrahepatic tumors, evaluating focal liver tumor therapy and vascular problems, and diagnosing solid organ injury, said Dr. Yuko Kono of the University of California, San Diego Medical Center.
"We need approval for radiologic applications of ultrasound contrast agents in the U.S.," she said.
Kono discussed UCSD's four years of experience with ultrasound contrast agents in radiologic applications during a presentation Tuesday at the Leading Edge in Diagnostic Ultrasound conference in Philadelphia. UCSD has performed more than 200 such studies under institutional review board (IRB) approval for off-label use.
Ultrasound contrast impacts patient care, she said. A UCSD research team evaluated 23 patients, who each received Optison (Amersham Health, Princeton, NJ), Definity (Bristol-Myers Squibb Medical Imaging, North Billerica, MA), or Imagent (Alliance Pharmaceutical, San Diego, CA) ultrasound contrast agents as recommended by a radiologist and/or ordered by a referring physician. Typically, the ultrasound contrast studies were requested due to an inconclusive imaging work-up.
Ultrasound was performed using phase-inversion real-time and intermittent imaging with a Sonoline Elegra scanner (Siemens Medical Solutions, Malvern, PA). Studies were performed before and after multiple contrast injections.
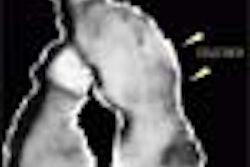
The organs and diseases studied included 15 liver lesions (seven hepatocellular carcinomas, two cholangiocarcinomas, one nodular regenerative hyperplasia, one regenerative nodule, one biliary infarction, one possible biliary problem, and two suspicious lesions) and six vascular lesions. One kidney mass and one spleen laceration were also included.
The ultrasound contrast study added important information in all 23 cases, and changed the diagnosis in seven of 23 cases (30%), Kono said. Patient management was changed in 11 of 23 cases (48%), and no adverse events were reported.
Monitoring HCC therapy
Kono also presented data specifically examining contrast-enhanced ultrasound’s utility for monitoring focal therapy of hepatocellular carcinoma (HCC). Previous research has shown CEUS to be useful in evaluating the efficacy of focal treatment for HCC, including RF ablation, ethanol ablation, and liver tumor therapy with chemoembolization (TACE).
In addition, UCSD researchers have shown previously that phase-inversion imaging is useful for evaluation of local HCC therapy and that CEUS was equal to MRI and angiography (and superior to CT) at assessing tumor viability following TACE, Kono said.
To investigate whether CEUS can predict treatment outcome earlier than CT or MRI (currently performed at one and two months after treatment), UCSD researchers studied 27 HCC patients scheduled for local therapy, including 32 TACE treatments, three RF ablation cases, and one ethanol ablation treatment.
CEUS exams were performed immediately before treatment in 25 cases, a few hours after in two cases, one day after in 11 cases, a week after in six cases, two weeks after in four cases, one month after in 11 cases, and more than one month after in 17 cases. Multiple bolus injections of Optison, Imagent, or Definity ultrasound contrast media were given intravenously, she said.
The patients received phase-inversion low-mechanical index (MI) real-time and intermittent imaging on an Elegra system at one frame per 1-5 seconds. A curved 3.5-MHz transducer at 1.6 to 2.5 MHz central transmit frequency was used, she said.
CEUS was compared to contrast-enhanced CT in 36 cases, MRI in 13, and x-ray angiography in 32 cases. Real-time imaging at low power allowed the detection of arterial blood flow in the viable portions of tumors, while intermittent imaging enabled the detection of persistent tumor parenchymal blood flow, she said.
In the pre-treatment studies, CEUS and CT and/or MRI all showed tumor enhancement, agreeing in all 15 cases, she said. In 26% (5 of 19) post-treatment studies, CT failed to detect persistent flow demonstrated by CEUS, she said. However, CEUS missed one tumor enhancement that was seen on MRI, and one that was visualized using CT.
In eight of 12 (67%) cases that received more than one CEUS after TACE, tumor vascularity remained visible following TACE, then decreased over time, she said. In four of the 12 cases, CEUS showed no tumor enhancement immediately post-treatment, with no flow detected over time.
"CEUS allows detection of persistent tumor blood flow," he said. "The effect of TACE on tumor flow increases over several days, and CEUS predicts outcome one to three days post-focal treatment."
By Erik L. RidleyAuntMinnie.com staff writer
May 14, 2003
Related Reading
Contrast-enhanced 3-D US boosts mammography results, April 11, 2003
Intramuscular gene transduction enhanced using microbubbles with ultrasound, February 25, 2003
Second-generation microbubbles enhance ultrasound's clinical utility, August 27, 2002
For prostate cancer screening, microbubbles are too big, and the options too few, July 5, 2002
Optison beats Levovist in US contrast face-off, March 6, 2002
Copyright © 2003 AuntMinnie.com