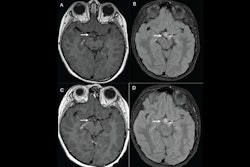
Bracco has secured U.S. Food and Drug Administration (FDA) approval to expand the use of its Vueway (gadopiclenol) contrast agent for MRI to neonates and infants.
Vueway is a macrocyclic gadolinium-based contrast agent (GBCA) that uses half the gadolinium dose as other macrocyclic GBCAs approved in the U.S., according to the company.
Guidance from radiology societies emphasizes the use of the lowest effective GBCA dose in contrast-enhanced imaging to reduce cumulative gadolinium exposure, Bracco added.
Vueway’s lower dose was designed to reduce exposure while maintaining diagnostic performance, a consideration the firm noted as particularly important in neonates and infants, whose brains and body tissues are still developing.
The FDA approval was based on data from a study that evaluated the safety and efficacy of Vueway injection in pediatric patients, according to Bracco.
Gadopiclenol was first approved by the FDA in September 2022 for adult patients and pediatric patients aged 2 and older. The EU approved an expanded indication covering neonates and infants in January 2026.