Researchers have introduced a novel MRI tumor marker called the “dark-light-dark sign” and suggest it can help predict lymph node metastasis in patients with rectal cancer, according to a study published December 23 in Radiology.
The sign is a three-layer pattern around tumors visualized by contrast-enhanced T1-weighted imaging, and it improved lymph node diagnosis when integrated into a multivariable prediction model and tested on a patient dataset, noted lead author Xin-Yue Yan, of Peking University Cancer Hospital and Institute in Beijing, China, and colleagues.
“Our two-center study demonstrated that the absence of the novel so-called dark-light-dark sign derived at contrast-enhanced T1-weighted imaging was associated with a higher risk of lymph node metastasis," the group wrote.
Lymph node metastasis (LNM) reduces five-year survival rates of patients with rectal cancer from 92% to 37%, and the presence or absence of LNM directly informs clinical management, the study authors explained. Pathologic evidence has confirmed that the risk of nodal metastasis in rectal cancer patients is associated with tumor invasion depth and volume, yet this clinical-pathologic association has yet to be translated into tumoral MRI feature-based predictors, they noted.
To bridge the gap, Yan and colleagues developed a predictive model that reflects this association. They defined the dark-light-dark sign as the presence of an inner hypointense tumor (dark), an intermediate hyperintense rim (light), and an outer hypointense muscularis propria, or muscle layer (dark). In brief, the absence of the sign indicates a more aggressive or later-stage tumor, the team noted.
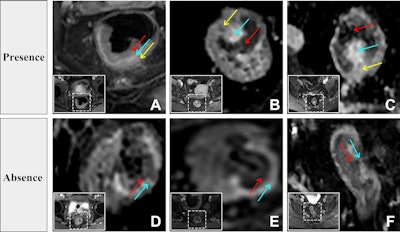 Evaluation of the dark-light-dark sign from contrast-enhanced T1-weighted images. The inset white solid and dashed boxes show the locations of the tumors. (A-C) Presence of the three-layered dark-light-dark sign, characterized sequentially by slight enhancement of the tumor (red arrow), obvious strip-shaped enhancement (blue arrow), and slight enhancement of the muscularis propria layer (yellow arrow). (D-F) Absence of the dark-light-dark sign. Only two layers are observed: the first dark layer (red arrow) and the light layer (blue arrow). The third dark layer -- slight enhancement of the muscularis propria layer -- is absent.RSNA
Evaluation of the dark-light-dark sign from contrast-enhanced T1-weighted images. The inset white solid and dashed boxes show the locations of the tumors. (A-C) Presence of the three-layered dark-light-dark sign, characterized sequentially by slight enhancement of the tumor (red arrow), obvious strip-shaped enhancement (blue arrow), and slight enhancement of the muscularis propria layer (yellow arrow). (D-F) Absence of the dark-light-dark sign. Only two layers are observed: the first dark layer (red arrow) and the light layer (blue arrow). The third dark layer -- slight enhancement of the muscularis propria layer -- is absent.RSNA
The researchers first trained the model using a dataset from 922 patients who underwent contrast-enhanced T1-weighted MRI scans prior to primary surgery between January 2016 and December 2024 at their center; 354 were LNM-positive, and 568 were LNM-negative. Next, they tested it on an external dataset from 149 patients -- 73 of whom were LNM-positive and 76 of whom were LNM-negative -- and compared its performance for predicting LMN to plain scan models, features recommended by the European Society of Gastrointestinal and Abdominal Radiology (ESGAR), and the lymph node short-axis diameter alone (tumor size).
According to the results, the enhanced model yielded an area under the receiver operating characteristic curve (AUC) of 0.89 in the external test set, outperforming the plain scan model (AUC, 0.79), ESGAR nodal status alone (AUC, 0.76), and lymph node short-axis diameter alone (AUC, 0.69) (all p < 0.001).
In addition, the enhanced model demonstrated superior diagnostic performance across lymph node size and pathologic T stage subgroups. In the training set, it significantly outperformed both the plain scan model and ESGAR criteria in all lymph node short-axis diameter subgroups, Yan et al reported.
“Integrating tumor characteristics -- especially the dark-light-dark sign -- with ESGAR status improved preoperative LNM diagnosis, particularly in patients with lymph nodes smaller than 5 mm or who had stage T2 tumors,” the study authors wrote.
Despite clinically meaningful AUC differences between the models, the external test set was small, and further research will be required to validate the model and determine its generalizability in larger cohorts, the researchers noted.
“This approach has promising potential for developing individualized risk-adapted strategies, particularly in patients with lymph nodes smaller than 5 mm and stage T2 disease,” they concluded.
The full study is available here.