A new brain imaging technique that combines 7-tesla MRI and arterial spin labeling (ASL) has shown how small blood vessels in the brain pulse with each heartbeat -- and could help clinicians better understand Alzheimer's disease, researchers have reported.
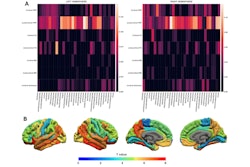
A group led by senior author Danny JJ Wang, PhD, of the University of Southern California (USC) used ultra-high field 7-tesla MRI to demonstrate how these microvessel pulses – called "microvascular volumetric pulsatility" – increase with age, especially in the brain's deep white matter, a region "critical for communication between brain networks," the university noted. The study findings were published September 25 in Nature Cardiovascular Research.
"Arterial pulsation is like the brain's natural pump, helping to move fluids and clear waste," Wang said. "Our new method allows us to see, for the first time in people, how the volumes of those tiny blood vessels change with aging and vascular risk factors. This opens new avenues for studying brain health, dementia, and small vessel disease."
As people age, their brains' white matter is susceptible to reduced blood supply of distal arteries, the blood vessels that carry blood away from the heart and into the farthest parts of the body, according to the group. Increasing microvessel pulses can disrupt systems in the brain, possibly speeding up memory loss and Alzheimer's disease, it explained.
"Excessive vascular pulsatility may impair the function of the brain's glymphatic system – a newly recognized network that clears waste products like beta-amyloid proteins that build up in Alzheimer’s disease," USC noted in its statement. "Over time, disrupted fluid circulation could accelerate cognitive decline."
Even though researchers have long understood that large artery stiffness and pulsatility are linked to increased risk of stroke, dementia, and small vessel disease, it has been difficult to measure these pulsations in the brain's smallest vessels without using invasive methods -- and usually only in animal studies.
Wang and colleagues combined the two techniques -- ASL and another called 4D vascular space occupancy (VASO) -- to track volume changes in microvessels over the cardiac cycle in 12 older individuals (median age, 60 years) and 11 younger ones (median age, 28). They reported that the older adults showed heightened microvascular pulsations in deep white matter compared to the younger individuals (p = 0.006), and that hypertension further intensified these changes.
"These findings provide a missing link between what we see in large vessel imaging and the microvascular damage we observe in aging and Alzheimer's disease," said lead author Fanhua Guo, PhD, in the USC statement. Guo is a postdoctoral researcher in Wang's lab.
The investigators plan to continue to explore how this type of imaging could be used more widely, including with 3-tesla MRI scanners, via future studies that will test whether "microvascular volumetric pulsatility predicts cognitive outcomes and whether it can serve as a biomarker for early intervention in Alzheimer's disease and related conditions."
"This [study] is just the beginning," Wang said. "Our goal is to bring this from research labs into clinical practice, where it could guide diagnosis, prevention, and treatment strategies for millions at risk of dementia."
The complete study can be found here.