Siemens Healthineers has received U.S. Food and Drug Administration (FDA) clearance for its Magnetom Terra.X 7-tesla MRI scanner.
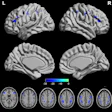
First showcased at RSNA 2022, Terra.X is a clinical 7-tesla system with eight-channel, parallel transmit architecture. It features what Siemens calls Ultra IQ technology – new hardware and software that has increased homogeneity and enabled a larger field of view, the company said. Ultra IQ is designed to exploit the resolution and contrast possible with 7-tesla scanning, according to the vendor.
Terra.X succeeds Siemens’ Terra clinical 7-tesla scanner, which was introduced in 2017. The FDA clearance also enables legacy Terra systems to be upgraded to Terra.X and be upgraded to Siemens’ syngo MR XA60A software.
Both Terra.X and Terra systems outfitted with MR XA60A software employ the company’s Deep Resolve AI-based image reconstruction technology, which has been trained specifically with 7-tesla data. Paired with accelerated acquisition techniques such as high parallel acquisition, Deep Resolve can complete a routine high-resolution brain and knee exam in under 20 minutes, Siemens said.
The company also highlighted the scanner’s new gradient system and its multinuclear option for clinical imaging of substances in the body such as sodium in the brain and phosphorus in muscle tissue.