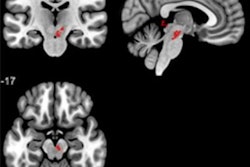
Ultrahigh-resolution MRI has identified enlarged perivascular spaces in the brains of people who suffer from migraines, research to be presented at the RSNA annual meeting in Chicago has found.
A team led by Wilson Xu from the University of Southern California, Los Angeles will highlight study results that showed significant differences in the perivascular spaces between people with migraines and those without migraines. Perivascular spaces are fluid-filled areas that surround blood vessels in the brain. They are affected by several factors, including abnormalities in the blood-brain barrier and inflammation.
"[Differences in perivascular spaces] might be suggestive of glymphatic disruption within the brain, but whether such changes affect migraine development or result from migraine is unknown," Xu and colleagues wrote in their abstract.
Over 37 million people in the U.S. are affected by migraines, and up to 148 million people worldwide suffer from the condition, according to figures from the American Migraine Foundation. Enlarged perivascular spaces can be a sign of underlying small vessel disease, but the potential ties between migraines and enlarged perivascular spaces have been unclear.
Xu's group used ultrahigh field 7T MRI to compare structural microvascular changes in people with different types of migraines and compared them to migraine-free controls. The team used data from 10 patients with chronic migraines, 10 with episodic migraines without aura, and five who were free of migraines.
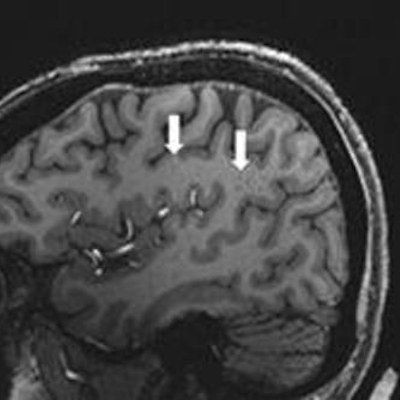
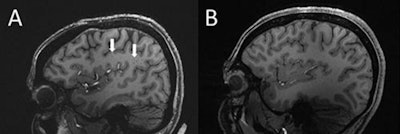 (A) Enlarged centrum semiovale perivascular spaces present (arrows) on sagittal T1-weighted MRI in a case with chronic migraine. (B) MRI shows a migraine-free control case without enlarged spaces. Image and caption courtesy of the RSNA.
(A) Enlarged centrum semiovale perivascular spaces present (arrows) on sagittal T1-weighted MRI in a case with chronic migraine. (B) MRI shows a migraine-free control case without enlarged spaces. Image and caption courtesy of the RSNA.They found that the number of enlarged perivascular spaces in the centrum semiovale was significantly higher with migraines compared to healthy controls (p = 0.04). The researchers also found that the frequency of white matter hyperintensities and cerebral microbleeds did not differ significantly between the migraine sufferers and the healthy control group. However, migraine patients showed a significant correlation between enlarged perivascular space in the centrum semiovale and the severity of deep white matter hyperintensities (p = 0.04).
The team noted that enlarged perivascular spaces may influence other vascular-related changes in people with migraines. This suggests that there could be a pathophysiological mechanism that could be used as an early imaging biomarker.
"Continued study with larger case populations and longitudinal follow-up will better establish the relationship between structural changes and migraine development and type," they concluded.