A single brain MRI and an artificial intelligence (AI) algorithm could effectively diagnose Alzheimer's disease (AD) -- even at an early stage, a study published June 20 in Communications Medicine has shown.
A team led by Dr. Marianna Inglese of the Imperial College London created an algorithm it calls an "Alzheimer's Predictive Vector" based on T1-weighted MRI data of brain shrinkage and found that it improved diagnostic accuracy compared to traditional methods of assessing hippocampal atrophy or cerebrospinal fluid beta amyloid.
The findings could translate into better care for patients, the authors noted.
"This method provides a biomarker able to detect an early stage of Alzheimer's Disease with a significant potential improvement of the clinical decision support system," they wrote.
Alzheimer's is the most common cause of dementia, and it can have a dramatic negative effect on memory, thinking, and behavior, the team explained. It's also often tricky to diagnose -- which may cause patients to suffer more than necessary. Techniques to identify Alzheimer's include memory and cognitive tests and brain scans that check for protein deposits in the brain and any shrinking in the hippocampus -- the part of the brain in charge of memory. Yet these tests take time to conduct and process.
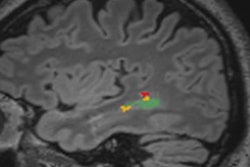
That's where MRI bolstered by AI could help, according to Inglese and colleagues. The group developed an algorithm for use with 1.5-tesla MRI -- a common device in most hospitals -- and trained it to identify and assess brain features like size, shape, and texture to predict Alzheimer's disease. The model utilizes established software for both brain segmentation and radiomics analysis (FreeSurfer); Inglese's group trained it with data taken from 783 people scanned with 1.5-tesla MRI.
Of the total patient cohort:
- 216 were healthy controls.
- 208 had mild cognitive impairment due to Alzheimer's.
- 181 had Alzheimer's.
- 94 had frontotemporal dementia.
- 84 had Parkinson's disease.
The investigators found that the algorithm identified subclinical brain shrinkage in those with early disease with better overall diagnostic accuracy compared to the techniques of assessing standard hippocampal atrophy or cerebrospinal fluid beta amyloid.
| Performance of AI algorithm compared to other ways of assessing for Alzheimer's disease | |||
| Measure | Standard hippocampal atrophy assessment | Cerebrospinal fluid beta-amyloid measurement | Alzheimer's predictive vector algorithm |
| Accuracy | 26% | 62% | 98% |
| Negative predictive value | 22% | 1% | 99% |
| Positive predictive value | 30% | 0.6% | 98% |
| Sensitivity | 29% | 1% | 98% |
| Specificity | 22% | 0% | 98% |
The findings suggest that a simple brain MRI exam could improve the diagnosis of Alzheimer's, said lead author Dr. Eric Aboagye, PhD, in a statement released by the college.
"Currently no other simple and widely available methods can predict Alzheimer's disease with this level of accuracy, so our research is an important step forward," he said.
This protocol could also make the diagnostic process less onerous for individuals and their loved ones.
"Waiting for a diagnosis can be a horrible experience for patients and their families," Aboagye said. "If we could cut down the amount of time they have to wait, make diagnosis a simpler process, and reduce some of the uncertainty, that would help a great deal. Our new approach could also identify early-stage patients for clinical trials of new drug treatments or lifestyle changes, which is currently very hard to do."