Quantitative MRI reveals brain structure abnormalities in premature babies that may develop into neurological conditions such as autism or cerebral palsy when the children are teenagers, according to a study published April 26 in Radiology.
The findings could help improve long-term outcomes in people born preterm (i.e., less than 28 weeks gestation), wrote a team led by Ryan McNaughton, a doctoral candidate in mechanical engineering at Boston University.
"Our results complement the existing body of evidence that abnormal developmental pathways [in preterm children] can be potentially understood with use of multiparametric quantitative MRI, with potential implications for improving diagnosis and therapeutic intervention," the group noted.
Although improvements in neonatal care have increased the survival rates of preterm babies, more research remains to be done to determine whether being born preterm increases risk of brain abnormalities later in life.
"So much of the maturation of the brain occurs during the third trimester when the fetus is in the womb's nourishing environment," said study co-author Dr. Thomas O'Shea of the University of North Carolina in Chapel Hill in a statement released by the RSNA. "These preterm babies don't experience that, so it seems likely that there are alterations in the brain maturation during that interval."
Twenty years ago, O'Shea and colleagues initiated a study called Extremely Low Gestational Age Newborn-Environmental Influences on Child Health Outcomes (ELGAN-ECHO) to examine effects of preterm birth. Over the years, the work has drawn in medical imaging experts such as study co-author and medical physicist Hernán Jara, PhD, also of Boston University.
For this current research, McNaughton's group used quantitative MRI to assess the brains of adolescents born preterm. The project included MRI data from 368 ELGAN-ECHO adolescents ages 14 to 16; of the total study cohort, 252 of the children developed neurotypically and 116 developed atypically. The investigators compared MRI results such as brain volume and proton density between the two cohorts and between boys and girls.
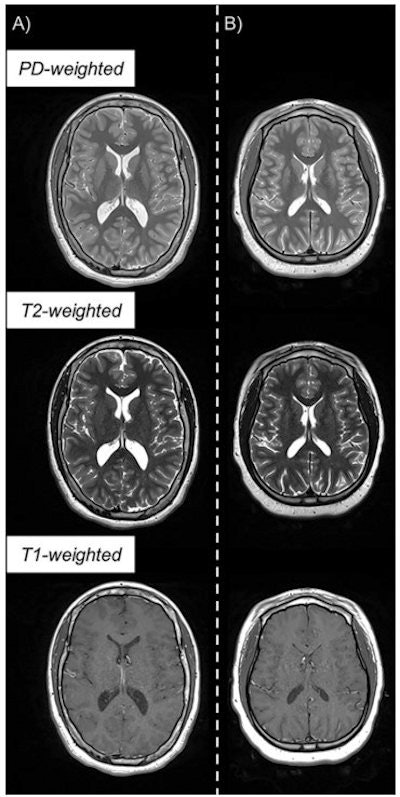 Example of triple turbo spin-echo brain MRI scans in (A) neurotypically developing and (B) atypically developing extremely preterm-born adolescents. Proton density (PD)-weighted, T2-weighted, and T1-weighted images are provided. MRI scans in the atypically developing participant (full-scale intelligence quotient < 85) demonstrated qualitatively smaller white and gray matter volumes and nearly indistinguishable tissue contrast compared with the neurotypically developing participant. Images and caption courtesy of the RSNA.
Example of triple turbo spin-echo brain MRI scans in (A) neurotypically developing and (B) atypically developing extremely preterm-born adolescents. Proton density (PD)-weighted, T2-weighted, and T1-weighted images are provided. MRI scans in the atypically developing participant (full-scale intelligence quotient < 85) demonstrated qualitatively smaller white and gray matter volumes and nearly indistinguishable tissue contrast compared with the neurotypically developing participant. Images and caption courtesy of the RSNA."Quantitative MRI in a large dataset allows you to identify small differences between populations that may reflect microstructural tissue abnormalities not visually observable from imaging," Jara said in the RSNA statement.
Those adolescents who had atypical neurological development showed this on quantitative MRI, with "subtle differences in white matter related to proton density that corresponded with less free water," the researchers noted. (Lower white-matter values are associated with atypical neurological development.)
| Quantitative MRI findings in white-matter volume in developing adolescents | ||||
| Volume | Neurotypically developing girls | Atypically developing girls | Neurotypically developing boys | Atypically developing boys |
| White-matter volume | 433 cm3 | 399 cm3 | 473 cm3 | 446c m3 |
At the beginning of the ELGAN-ECHO study, researchers collected umbilical and blood samples from the preterm infant participants. Going forward, McNaughton's team plans to use data from these samples to explore if there are connections between the quantitative MRI findings and the presence of toxins such as cadmium, arsenic, or other metals, and to evaluate the quantity and quality of myelin, a protective nerve covering crucial to healthy cognitive development.
"Further studies are needed to complement these findings with nonimaging clinical outcomes and perinatal inflammatory antecedents," the investigators concluded.