MRI scans show that accumulated iron in the outer layer of the brain is linked to cognitive deterioration in people with Alzheimer's disease, according to a study published June 30 in Radiology.
The results suggest that tracking iron accumulations in the brain could help clinicians better assess the scope and progression of disease, study co-author Dr. Reinhold Schmidt of the Medical University of Graz in Austria said in a statement released by the RSNA.
"We found indications of higher iron deposition in the deep gray matter and total neocortex, and regionally in temporal and occipital lobes, in Alzheimer's disease patients compared with age-matched healthy individuals," he said.
Previous studies have linked high levels of brain iron with Alzheimer's disease, wrote a team led by Dr. Anna Damulina, also of the university: Iron deposits are associated with beta amyloid, a protein that forms plaques between neurons and disrupts brain function, as well as tangles of another protein called tau that also collects inside neurons. But these studies have focused on the deep gray-matter structures of patients with Alzheimer's disease rather than the outer layer of the brain called the neocortex.
Unfortunately, the neocortex can be challenging to image with MRI, since the area's anatomy may make MRI susceptible to distortions, according to Damulina and colleagues. The best way to address this limitation is to use ultrahigh-resolution scans, but these can take too much time in the clinical setting.
So the researchers assessed baseline levels of brain iron in 100 patients with Alzheimer's disease and 100 healthy controls using a more efficient combination of a 3-tesla MR scanner and a technique called R2* mapping, which detects increased iron concentrations in the body (higher values translate to higher levels of iron).
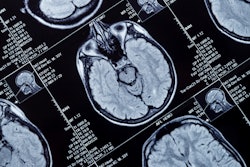
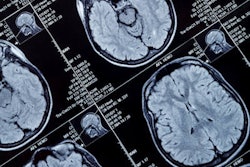
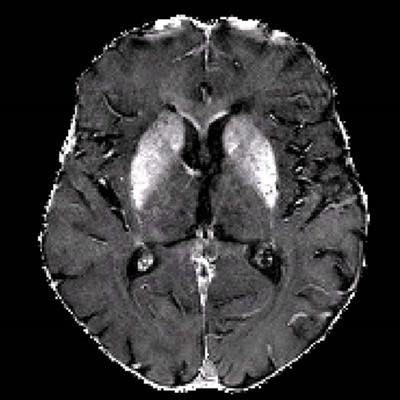
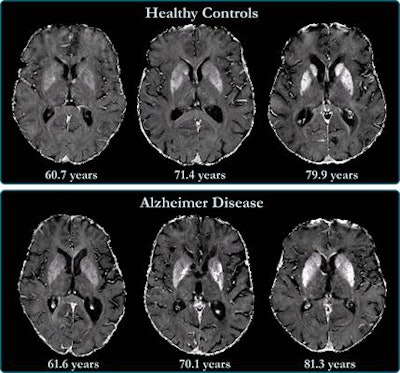 R2* maps of healthy control participants and participants with Alzheimer disease. R2* maps are windowed between 10 and 50 sec-1. Images courtesy of the RSNA.
R2* maps of healthy control participants and participants with Alzheimer disease. R2* maps are windowed between 10 and 50 sec-1. Images courtesy of the RSNA.The study found higher iron deposits in the Alzheimer's patients' deep gray matter, total neocortex, and in the temporal and occipital lobes compared to healthy controls.
| Comparison of median R2* levels in healthy controls and Alzheimer's patients | ||
| Area of the brain | Healthy controls | Alzheimer's patients |
| Median R2* levels | ||
| Basal ganglia | 29 sec-1 | 30.2 sec-1 |
| Total neocortex | 17 sec-1 | 17.4 sec-1 |
| Occipital | 19.6 sec-1 | 20.2 sec-1 |
| Temporal | 16.4 sec-1 | 18.1 sec-1 |
The study results suggest that it would be beneficial to further explore the use of chelators, drugs that reduce iron in the brain, in Alzheimer's treatment.
"Our study provides support for the hypothesis of impaired iron homeostasis in Alzheimer's disease and indicates that the use of iron chelators in clinical trials might be a promising treatment target," Schmidt said in the statement. "MRI-based iron mapping could be used as a biomarker for Alzheimer's disease prediction and as a tool to monitor treatment response in therapeutic studies."