What is the best way to overcome artifacts in patients wearing metallic orthodontic devices? The authors of a new study published November 12 in Radiology recommend slight modifications in functional MRI (fMRI) and diffusion-tensor MR imaging (DTI-MRI) sequences to complete the task.
Researchers from Johns Hopkins University took a cue from a previous study that used T2-prepared blood oxygenation level-dependent (BOLD) fMRI and a diffusion-prepared DTI sequence to work around the negative effects of metal objects, such as dental braces. Both approaches provided better signal- and contrast-to-noise ratios to bypass artifacts and create usable images.
"Susceptibility artifacts caused by metallic objects at echo-planar imaging (EPI) have been a major limitation for functional MRI and diffusion-tensor imaging in participants wearing metallic orthodontic material and other head implants," wrote the authors, led by Xinyuan Miao, PhD, from Johns Hopkins' division of MRI research. "T2-prepared BOLD functional MRI and diffusion-prepared DTI showed preserved signal-to-noise ratio (SNR) in the entire brain, whereas conventional EPI approaches showed significantly reduced SNR in regions with strong susceptibility effects."
As more children and adults are fitted for metal braces and other oral devices, MRI scans have become more challenging. Echo-planar imaging, which is needed to perform fMRI and DTI, is particularly susceptible to significant artifacts, such as signal reduction and image distortion, from these devices, the authors noted.
To work around this deficiency, researchers of a previous study reported success in using a whole-brain T2-prepared BOLD fMRI approach and a diffusion-prepared DTI sequence, followed by 3D fast gradient-recalled echo (GRE) sequence. The T2-prepared BOLD fMRI "showed little susceptibility artifacts throughout the brain and greater functional sensitivity than GRE EPI BOLD fMRI in regions near air-filled cavities in healthy participants and around the lesions containing blood products in presurgical patients," Miao and colleagues wrote of the previous research.
To find out if this same approach could work with metal orthodontic devices, the researchers prospectively performed 3-tesla MRI scans (Ingenia, Philips Healthcare) on six healthy subjects (mean age, 40 ± 6 years). Each exam included the following five protocols:
- Magnetization-prepared rapid acquisition gradient echo (MP-RAGE)
- 2D GRE EPI BOLD fMRI
- 3D T2-prepared BOLD fMRI
- 2D spin-echo EPI DTI
- 3D diffusion-prepared DTI with 3D fast GRE
In analyzing the results, Miao and colleagues found that T2-prepared BOLD fMRI and 3D diffusion-prepared DTI with 3D fast GRE both outperformed EPI for signal-to-noise ratio in the entire brain, which resulted in much better image quality. T2-prepared BOLD fMRI also significantly bested conventional EPI in contrast-to-noise ratio in regions with strong susceptibility to metal braces.
Since a picture is worth a thousand words, the researchers also observed much better results with diffusion-prepared DTI with 3D fast GRE, compared with spin-echo EPI DTI.
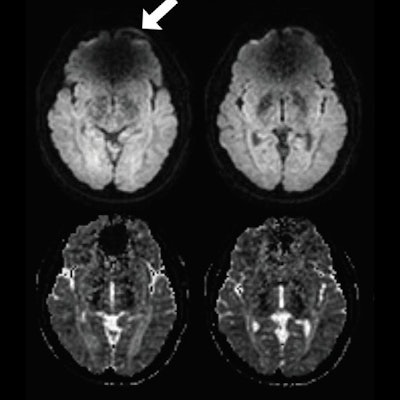
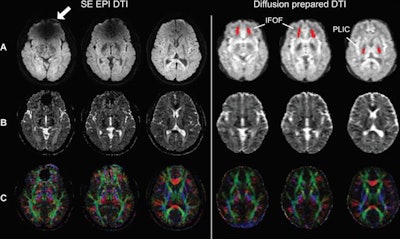 Spin-echo (SE) echo-planar imaging (EPI) and diffusion-prepared diffusion tensor imaging (DTI) axial images acquired at 3 tesla in a subject with metal braces. Shown as raw diffusion-weighted images (A), calculated apparent diffusion coefficient maps (B), and a color-coded fractional anisotropy map (C). Susceptibility artifacts were observed on SE EPI in regions close to braces (arrow). No obvious artifacts were seen on diffusion-prepared DTI. Regions of interest also are indicated in the inferior fronto-occipital fasciculus (IFOF) and posterior limb of internal capsule (PLIC) for quantitative analysis diffusion-prepared DTI (red). Images courtesy of Radiology.
Spin-echo (SE) echo-planar imaging (EPI) and diffusion-prepared diffusion tensor imaging (DTI) axial images acquired at 3 tesla in a subject with metal braces. Shown as raw diffusion-weighted images (A), calculated apparent diffusion coefficient maps (B), and a color-coded fractional anisotropy map (C). Susceptibility artifacts were observed on SE EPI in regions close to braces (arrow). No obvious artifacts were seen on diffusion-prepared DTI. Regions of interest also are indicated in the inferior fronto-occipital fasciculus (IFOF) and posterior limb of internal capsule (PLIC) for quantitative analysis diffusion-prepared DTI (red). Images courtesy of Radiology."Our results showed that T2-prepared blood oxygenation level-dependent functional MRI and diffusion-prepared diffusion tensor imaging can be useful alternatives for human brain mapping when echo-planar imaging fails to provide robust signals in the presence of metallic implants such as dental braces," Miao and colleagues concluded.