A new gadolinium-based contrast material could form the basis for submillimeter detection of tumors on MRI scans and provide a warning of early-stage liver disease sooner than conventional MRI contrast agents, according to findings from an animal study released October 29 in Nature Communications.
Researchers from Georgia State University are highlighting results they achieved testing the agent, which they believe could prove safer and more effective than existing gadolinium-based contrast agents (GBCAs). The new material could enable the visualization of liver tumors as small as 0.1 mm to 0.2 mm on MRI scans, while requiring less gadolinium than existing GBCAs.
The researchers noted that nonalcoholic fatty liver disease and alcoholic liver disease are common causes of chronic liver disease worldwide, with deaths from liver disease in the U.S. growing 31% from 2010 to 2015 among people ages 45 to 64. While liver disease is a slow-moving process, it can progress to become serious if it's not detected early, according to a team led by Jenny J. Yang, PhD, of Georgia State University.
While MRI has advantages over other imaging modalities for detecting nonalcoholic steatohepatitis and hepatic fibrosis caused by liver disease, it faces challenges in detecting the disease in its earliest stages.
To address the problem, Yang founded InLighta BioSciences, a spinoff of the university that received $2 million grant in 2017 from the U.S. National Cancer Institute to commercialize a protein-based contrast agent, ProCA32.collagen1, that targets overexpression of the biomarker collagen. Yang and colleagues describe how the gadolinium-based agent works in their new article in Nature Communications.
In the study, researchers tested ProCA32.collagen1 in mice. The mice received injections and were put on a diet that promoted early-stage liver fibrosis. They were then scanned after injection of the agent at 1.4- and 7.0-tesla field strengths.
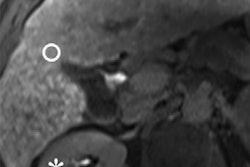
The study showed that ProCA32.collagen1 was able to differentiate fibrotic regions from healthy background tissue. If used in humans, this could help overcome the limitations of invasive biopsies that can't analyze the entire liver.
What's more, the researchers did not detect any gadolinium deposition in the brains of the mice, a finding that they said indicated that ProCA32.collagen1 performed equivalently to macrocyclic gadolinium agents. Additionally, ProCA32.collagen1 can be used at much lower doses than other GBCAs, reducing the risks of gadolinium toxicity.
"The contrast agent is expected to overcome the major clinical barriers in early diagnosis, noninvasive detection, and staging of chronic liver diseases and have strong translational potential for monitoring treatment efficacy," Yang and colleagues concluded.