A new study published online July 28 in Radiology has disturbing news about gadolinium contrast retention. Colorado researchers found that pediatric patients given a linear gadolinium-based contrast agent (GBCA) had increased MR signal intensity in a certain region of the brain.
Young patients who received the gadolinium contrast agent showed greater signal intensity in the dentate nucleus on unenhanced T1-weighted MR images, compared with children who never received the contrast agent. The findings are of particular concern because pediatric brains are still developing, and these children may be exposed to additional gadolinium in future scans, depending on their illness.
 Dr. Thomas Flood from the University of Colorado.
Dr. Thomas Flood from the University of Colorado."There is indirect evidence that this change in signal intensity is related to gadolinium deposition in the dentate nucleus, which is in line with adult data," said lead author and third-year radiology resident Dr. Thomas Flood from the University of Colorado Children's Hospital.
Gadolinium controversy
In recent years, studies have surfaced confirming traces of gadolinium in the brain tissue of deceased adults who had normal renal function many years after their contrast-enhanced MRI scans. There is, however, scarce research into gadolinium exposure among pediatric patients.
To date, there is no known definitive risk associated with deposition of gadolinium from the levels used in GBCAs. What is known is that gadolinium can be toxic, can cause adverse allergic reactions, and has been linked to nephrogenic systemic fibrosis (NSF) in adult patients with insufficient renal function.
"At the University of Colorado Children's Hospital, we have a large pediatric patient population that has contrast-enhanced [MRI] exams," Flood told AuntMinnie.com. "So we decided to see if this effect is replicated in pediatric patients. We assumed it would be, but we didn't know if it was actually happening, to what extent, or if there are any important differences."
Pediatric patients also pose unique challenges when clinicians have to decide whether a GBCA-enhanced MRI scan is warranted. One question is what effect gadolinium deposition could have on a child's still-developing brain.
"Some patients in our study were scanned with contrast-enhanced exams at less than 1 year of age," Flood said. "Depending on their medical issue or history, they potentially could have multiple repeat contrast-enhanced scans in their life. The amount of gadolinium they are exposed to could potentially be greater than an adult who starts getting exams when they are at middle age or older."
GBCA comparisons
The retrospective study included 46 pediatric patients who had undergone at least three MRI scans with gadopentetate dimeglumine (Magnevist, Bayer HealthCare) and 57 age-matched control subjects who had never received a GBCA. Subjects ranged in age from older than 31 days to younger than 18 years, were scanned between November 2005 and July 2013, and had normal brain MRI results on their most recent report (Radiology, July 28, 2016).
All subjects were imaged on a 1.5-tesla whole-body MRI scanner (Avanto, Siemens Healthineers), which included an unenhanced axial T1-weighted spin-echo sequence. Measurements of signal intensity were taken within the globus pallidus, thalamus, dentate nucleus, and pons, with a mean diameter of 5 mm (range: 3-9 mm).
Flood and colleagues discovered higher signal intensity in the dentate nucleus among patients who previously received multiple GBCA-enhanced exams (mean ratio, 1.046 ± 0.006), compared with subjects who had not received contrast (mean ratio, 1.007 ± 0.0058). This result is similar to what has been confirmed in adult patients.
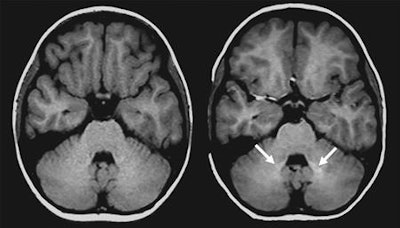 Axial unenhanced T1-weighted spin-echo brain MRI shows that dentate nucleus signal intensity increases with repeated exposure to linear GBCA. Left scan is of a 6-year-old girl before contrast material exposure; right scan is of the same patient at 9 years of age after eight GBCA-enhanced scans. Arrows indicate increased bilateral signal intensity in the dentate nucleus. Image courtesy of Radiology.
Axial unenhanced T1-weighted spin-echo brain MRI shows that dentate nucleus signal intensity increases with repeated exposure to linear GBCA. Left scan is of a 6-year-old girl before contrast material exposure; right scan is of the same patient at 9 years of age after eight GBCA-enhanced scans. Arrows indicate increased bilateral signal intensity in the dentate nucleus. Image courtesy of Radiology.However, there was no such signal intensity increase in the globus pallidus in the GBCA-exposed group (mean ratio, 1.014 ± 0.0091), compared with the GBCA-free subjects (mean ratio, 1.131 ± 0.0070).
"This is where the pediatric study's findings differ from the adults, where gadolinium deposition was found in the globus pallidus," Flood said. "We think most likely pediatric patients may not have had much gadolinium exposure; if they had, we would see more [deposition] in the globus pallidus."
The researchers also found no statistically significant correlation in the mean time between GBCA-enhanced MRI scans and the unenhanced T1 signal intensity measurements taken of the dentate nucleus.
"However, we did see a dose-dependent increase in dentate nucleus signal intensity with total cumulative gadolinium exposure," Flood added. "Taken together, these two findings suggest that gadolinium is depositing in the dentate nucleus and staying there, without significant clearance over time."
Follow-up research
The "clinical significance of our findings is unknown," the researchers wrote, adding that further studies with larger patient cohorts with different GBCAs are needed to confirm the presence and effects of gadolinium on pediatric patients.
"Since there is no known pathology from intracranial deposition of gadolinium, but there is potential risk, our group suggests the benefits of contrast-enhanced exams should be weighed against the potential risks before administering a linear gadolinium-based contrast agent to children," Flood said.
He and his colleagues are continuing their research by looking into how pediatric patients are reacting to the facility's current use of gadobenate (MultiHance, Bracco) for MRI scans. Like Magnevist, MultiHance a linear ionic agent. (Flood did not know why the hospital changed GBCAs.)
They also plan to investigate the GBCA issue with postmortem tissue analysis, as was performed in past studies in adults, to confirm whether traces of gadolinium are accumulating in the brain tissue.
"The most interesting question is whether this causes any clinical harm at the microscopic level or higher, but that will require additional novel investigations," Flood said.