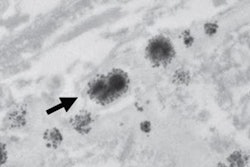
Researchers from the Mayo Clinic can't quite explain the clinical significance, but the discovery of traces of gadolinium in four areas of the brain several years after administration of MRI contrast is enough evidence to warrant further investigation as to the reasons why.
The researchers found concentrations of gadolinium in the neuronal tissues of the dentate nuclei, pons, globus pallidus, and thalamus in autopsies of 13 deceased patients who received gadolinium-based contrast agents (GBCAs) between 2000 and 2014. The study, published online March 5 in Radiology, recorded between 0.1 and 58.8 micrograms of gadolinium per gram of tissue in the four brain regions of the patients, who had relatively normal renal function at the time of their MRI exams.
Lead author Dr. Robert McDonald, PhD, and colleagues from the Mayo Clinic in Rochester, MN, noted that deposits appear to occur in all patients exposed to gadolinium and were detectable with as few as four doses of GBCA.
"Additional studies are needed to investigate the clinical significance of these findings and the generalizability to other GBCAs," they wrote.
Gadolinium utilization
The development of gadolinium-based contrast has greatly expanded the diagnostic capabilities of MRI, with previous studies estimating the annual number of administered doses of GBCAs at more than 10 million.
However, the use of gadolinium contrast took a hit in 2006 due to a possible connection between gadolinium and nephrogenic systemic fibrosis (NSF), a debilitating affliction characterized by widespread tissue fibrosis, in dialysis patients and those with insufficient renal function.
Heightened awareness of NSF and strict MRI protocols resulted in a significant decrease in the number of adverse events from gadolinium, and subsequent studies confirmed that the chance of experiencing NSF or other adverse reactions for otherwise normal patients was generally less than 0.1%.
More recently, however, several studies have noted increases in T1-weighted MRI signal in various central nervous system (CNS) structures following repeated gadolinium administration in patients with normal renal function, McDonald and colleagues wrote.
Therefore, they sought to validate these findings of CNS gadolinium deposition with direct assessment of gadolinium accumulation in neuronal tissues among deceased patients previously exposed to multiple doses of intravenous gadolinium.
Autopsy samples
The researchers took tissue samples from 23 deceased patients: 13 had received MRI contrast and 10 were used as a control group. The contrast group had undergone four to 29 contrast-enhanced MRI scans, whereas the control group had received one to six unenhanced MR exams between 2000 and 2014.
The contrast group had a median age of 51 years at the time of death, compared with a median age of 83.5 years for the control group. Median age at the time of the first MRI scan was 51 years for the contrast group, compared with 78.5 years for the control group.
McDonald and colleagues imaged the deceased patients' entire brains using 1.5-tesla MRI, with an unenhanced T1-weighted axial sequence. They also assessed the subjects' renal function before each MR exam by using the estimated glomerular filtration rate (eGFR) derived from serum creatinine results taken within 24 hours of MRI.
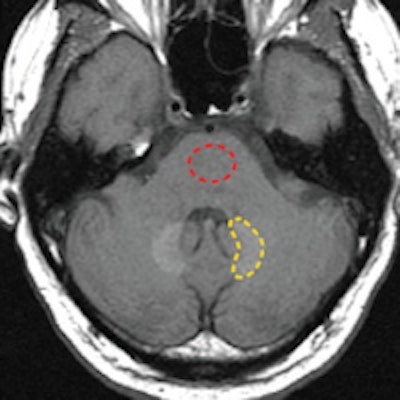
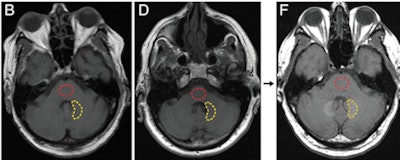 Axial T1-weighted MR images show the posterior fossa at the level of the dentate nucleus. Image B is of a control group patient, while image D is the first MRI exam and F is the last MRI exam of a patient in the contrast group. Regions of interest used to quantify signal intensity are shown as dashed lines for the dentate nucleus (yellow) and pons (red). Images courtesy of Radiology.
Axial T1-weighted MR images show the posterior fossa at the level of the dentate nucleus. Image B is of a control group patient, while image D is the first MRI exam and F is the last MRI exam of a patient in the contrast group. Regions of interest used to quantify signal intensity are shown as dashed lines for the dentate nucleus (yellow) and pons (red). Images courtesy of Radiology.Gadolinium discoveries
As expected, the researchers found no detectable levels of gadolinium among the control subjects. However, there were elevated levels of gadolinium in four regions of the brain -- the dentate nucleus, pons, globus pallidus, and thalamus -- in the contrast subjects, ranging from 0.1 to 58.8 micrograms of gadolinium per gram of tissue.
The dentate nucleus contained the highest median concentration of gadolinium (6.6 micrograms per gram of tissue), while the pons had the lowest median concentration (0.3 micrograms per gram of tissue). The globus pallidus had 1.7 micrograms and the thalamus had 0.5 micrograms, the researchers found.
There were also moderate to strong correlations between T1-weighted MR signal intensity changes and tissue concentrations of gadolinium in all four brain regions, according to the group. This ranged from a Spearman rank correlation coefficient of 0.49 (p = 0.08) in the globus pallidus to 0.89 (p < 0.0001) in the dentate nucleus.
"Our findings suggest that intravenous administration of GBCA is associated with dose-dependent deposition in neuronal tissues that is unrelated to renal function, age, or interval between exposure and death," the authors wrote.
However, they also conceded that the "clinical significance of these findings is incompletely understood at this time." The results "strongly argue for future research to assess the in vivo stability and safety of GBCAs," they concluded.