Taking cardiac MR images and turning them into 3D prints of the entire heart has gone from experiment to clinical use in Peoria, IL. It is helping doctors there plan complex congenital heart surgeries in young children, according to a November 19 presentation at the American Heart Association (AHA) annual meeting in Chicago.
Dr. Matthew Bramlet discussed his group's technique for creating life-sized plaster hearts from MR images of three young patients who needed complex cardiac surgery. He spoke with AuntMinnie.com about the work in advance of his talk.
 A printed 3D heart is marked to show anatomy. All images courtesy of Dr. Matthew Bramlet.
A printed 3D heart is marked to show anatomy. All images courtesy of Dr. Matthew Bramlet.Heart surgeons still rely mostly on 2D images acquired using angiography, echocardiography, and MRI for surgical planning, but such scans can be inadequate in many cases, he said.
"We use a ton of ultrasound and cardiac cath ... but for complex cases, MRI is our primary imaging modality," said Bramlet, a cardiologist at the Children's Hospital of Illinois and an assistant professor of pediatrics at the University of Illinois College of Medicine.
The 3D printed heart project at the Jump Trading Simulation and Education Center and the Children's Hospital of Illinois (both affiliated with OSF HealthCare and the University of Illinois College of Medicine) treats about 200 young patients a year who have congenital heart defects. For most of them, the reconstructed MR images are sufficient, but a handful of cases are so complex that the surgeons hesitate to approach them without a real 3D model.
"My job as a pediatric cardiologist is to provide the best information to our surgeon, so when he goes in he doesn't have any surprises," Bramlet said.
Patients born with just half a heart require a series of surgeries to reroute the blood flow through the heart without disrupting the function of the various components.
"In some instances, that is a very complex 3D pathway the surgeon has to create -- essentially on the fly," he said. "They make the best decisions they can, but in some cases the complete understanding of the situation isn't realized until they are in the operating room."
 Dr. Matthew Bramlet and a colleague review cardiac CT images.
Dr. Matthew Bramlet and a colleague review cardiac CT images.At the 3D heart project, which was initiated two years ago, 3D printing is beginning to end the nerve-wracking mystery of complex spatial orientation problems that surgeons might encounter during operations -- to the benefit of them and their patients.
"That is what we can offer, and that, to me, is the tremendous potential of this," Bramlet said. 3D printing is the only way he knows of navigating the intricate and complex pathways that are best understood before the patient gets on the table, he added.
Image acquisition and analysis
The process begins with acquisition of the MR images on a 1.5-tesla scanner (Signa HDxt 1.5T, GE Healthcare), followed by automated analysis of the DICOM MRI datasets using software (Mimics Innovation Suite 16.0, Materialise).
The software processing enables reconstruction of a 3D rendered model that can communicate with the 3D printer (ZPrinter 450, Z Corporation). The Mimics software employs a thresholding tool to highlight portions of the cardiac MR images that would be reproduced in the model in three imaging planes.
To build the model in a logical way that allowed for a complete intracardiac anatomic assessment of the three cases in the study, the process began with image sequences focused on intracardiac detail rather than blood pool for accurate reproduction of cardiac structures, the authors explained in an abstract.
"In the volume-rendered images, the contrast injection makes the blood pool visible, and you can rotate the aorta and see this contrast-enhanced blood," which is a fine approach for extracardiac structures such as veins and the aorta, Bramlet said. Unfortunately, this technique is of limited utility when you're trying to figure out intracardiac anatomy, the location of the holes in the heart, and how to reconfigure the pumping chambers to make them functional.
"So from the beginning we started using high-resolution imaging rather than contrast blood-pool imaging to accurately recreate the heart," Bramlet said. "It's hard, and a couple of things have to happen. It's garbage in, garbage out, so you have to have good images to start with."
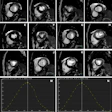
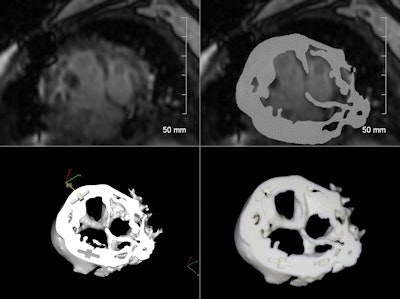 Cardiac MR images before (top) and after (bottom) reconstruction in preparation for 3D printing.
Cardiac MR images before (top) and after (bottom) reconstruction in preparation for 3D printing.The MR images are created using a 3D HEART sequence employing a 3D respiratory-navigated balanced steady-state free precession (SSFP) multislab sequence with T2 preparation that provides whole-heart coverage with a high contrast-to-noise ratio between vessels and myocardium, according to the group.
The model images are then reconstructed similar to a short-axis stack with three components (the basal portion, a middle slice containing all four valves, and the apical portion) for improved orientation. This method creates an accurate 3D printed representation of the patient's anatomy with slices set in a familiar orientation and localizing tabs for easy manipulation in the surgeon's hands.
In all three cases discussed, the model delivered key information that wasn't available from traditional imaging. The information improved the surgeon's preoperative understanding of the anatomy being studied, altering the surgical plan, Bramlet said.
In one case, for example, the 3D heart model showed a "Swiss cheese-type" ventricular septal defect that wasn't seen on other imaging modalities, including preoperative transesophageal echocardiography. Use of the heart model changed the surgical plan and shortened the cross-clamp time, the authors wrote in an abstract.
In the second case, complex intracardiac relationships shown in the 3D model changed the surgical plan from a single ventricle repair to a successful two-ventricle repair.
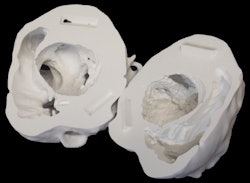 A 3D printed heart is split in half to reveal its internal structure.
A 3D printed heart is split in half to reveal its internal structure."Imagine a scenario where you have four chambers, but the hole between two of them is in a remote location compared to where it needs to be," Bramlet said. "The hole is usually between the right and left ventricles. Depending on the anatomy, if the hole is in a good spot, you can just patch across the hole ... but if the hole is in a bad spot, you can't patch all this real estate; you have to have a direct route, and in the second case that's what we did."
In the third case, the 3D model delineated the relationship between an aneurysm and the left coronary artery, again altering the surgical approach.
"The 3D printing made it so obvious," Bramlet said. "You're creating a higher level of understanding. Then, when you go back and look at the 2D images, you can even understand those better. It augments the understanding to such a degree that some of the surgeons won't do these complex cases until they have a heart in the hand now."
Slice-by-slice review
Of course, the job isn't done there. Before the model can be used in surgical planning, Bramlet has to go back to the images, slice by slice, to ensure that what's in the model is actually part of the anatomy and not, for example, blood that the software has mistaken for tissue.
"If we're making medical decisions based on this model, we have to make sure the model is an exact representation of the anatomy," he said. "What you're looking at through the model is a human interpretation of images through a computer program."
Interestingly, though, at a recent 3D printing forum, the focus was on whether the model was a precise replica of the images, rather than whether the model was a perfect copy of the anatomy, Bramlet said. The uncertainty comes from the soft-tissue aspect of cardiac model building, as 3D printing's origins and reputation are in bone replication.
"I have an incredible group of engineers who do a majority of the work, and then I come in and sort of clean it up," said Bramlet, who is trained in cardiac imaging. "I look at each slice and I make sure the marks sitting on top of each slice actually represent the anatomy, and that requires an understanding of images, and of the modality's limitations and strengths. The process that goes into 3D printing from cardiac images is perhaps the most critical step in creating a model that mimics material life."