Canadian intraoperative MRI technology developer IMRIS has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its IMRISNV and IMRIScardio products.
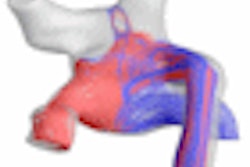
IMRISNV features a wide-bore, 3-tesla MRI scanner and a biplane angiography system specifically designed to coexist in the same suite; new MR images can be acquired to assess the effectiveness of treatment before, during, and after procedures. IMRIScardio features a wide-bore, 1.5-tesla MRI scanner that helps physicians visualize the cardiovascular system before, during, and after an intervention, according to the Winnipeg, Manitoba-based firm.
Both systems are available in multiple room configurations to provide diagnostic, interventional, and surgical capabilities, IMRIS said.
Related Reading
IMRIS wins CE Mark for MR angiography suite, May 1, 2009
IMRIS lands China install, April 16, 2009
IMRIS launches research initiative, February 20, 2009
IMRIS signs Yale, January 13, 2009
IMRIS takes first Australian order, January 7, 2009
Copyright © 2009 AuntMinnie.com