Genicular artery embolization can improve pain and knee function for at least three months in patients with knee osteoarthritis, according to a study published on December 15 in Diagnostic and Interventional Imaging.
The finding comes from a first-in-human trial of genicular artery embolization (GAE) using an ethiodized oil-based emulsion for the temporary treatment of knee osteoarthritis, and it suggests that the technique is safe for pain management in patients who are not candidates for surgery, the researchers noted.
“GAE using an ethiodized oil-based emulsion is safe and improves pain and function in participants with [knee osteoarthritis] for at least three months,” wrote corresponding author Marc Sapoval, MD, PhD, chair of cardiovascular radiology at Hôpital Européen Georges-Pompidou in Paris.
GAE is an emerging minimally invasive intervention for patients with painful knee osteoarthritis who are refractory to other treatments or who are reluctant to undergo or ineligible for knee replacement surgery, the researchers explained.
The imaging-guided procedure involves inserting microparticles to target genicular arteries that supply the synovial lining of the knee. These microparticles block blood flow, which reduces pain signals and provides relief, they wrote. Since the introduction of GAE, however, studies demonstrating its effectiveness have only used permanent microparticles, with permanent embolization carrying a risk of ischemic skin or bone damage, they noted.
In this trial, the group for the first time tested the use of a temporary ethiodized (iodine treated) oil-based embolic agent for GAE whose embolizing properties moreover have already been established in previous studies. Specifically, the author noted that in unpublished personal data, they confirmed the agent was able to temporarily embolize knee arteries for 10 minutes.
The group enrolled 22 consecutive participants (13 women, mean age: 66 years old). The emulsion for the GAE procedures consisted of a mixture of ioversol and ethiodized oil (ratio 1-to-3) prepared extemporaneously. Efficacy was assessed based on patient-reported changes in pain using visual analog scale (VAS) scores, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) function scores, and outcome measures in rheumatoid arthritis clinical trials (OMERACT).
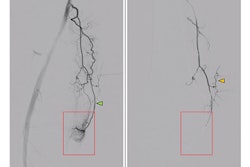
 Angiograms of the right knee in a 76-year-old man with knee osteoarthritis. A) Before embolization, selective angiography of the common trunk of the superior lateral and superior medial genicular arteries shows hypervascular inflammatory blush (arrows). B) During embolization using ethiodized oil of the common trunk, the angiogram shows emulsion droplets (arrows). Image courtesy of Diagnostic and Interventional Imaging.
Angiograms of the right knee in a 76-year-old man with knee osteoarthritis. A) Before embolization, selective angiography of the common trunk of the superior lateral and superior medial genicular arteries shows hypervascular inflammatory blush (arrows). B) During embolization using ethiodized oil of the common trunk, the angiogram shows emulsion droplets (arrows). Image courtesy of Diagnostic and Interventional Imaging.
According to the findings, the immediate technical success rate was 100%. The rate of serious adverse events within one month was 5% (1/22) were and related to reversible worsening of renal function.
In terms of patient pain, highlights included the following:
- The mean VAS pain score dropped from 74.4 mm at baseline to 37.2 mm at three months (p < 0.001).
- The mean WOMAC function score (with scoring ranging from 0 to 100) decreased from 57.3 at baseline to 33.5 at three months (p < 0.001).
- At three months, 16 of 22 participants (73%) were considered responders according to the OMERACT responder criteria, including high improvement in either pain or WOMAC function, or improvement in both pain and WOMAC function.
Ultimately, there is an unmet need for minimally invasive techniques that allow conservative management of knee osteoarthritis patients in whom total or partial knee replacement is not indicated, the authors wrote.
“If our results are confirmed in larger, randomized, controlled trials, ethiodized oil-based emulsion could become one option for GAE,” the researchers concluded.
The full study can be found here.