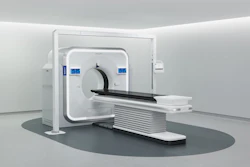
Philips has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Philips Spectral CT Verida system.
Verida is an AI-powered spectral CT system that features a third-generation dual-layer detector and deep-learning reconstruction engine.
It incorporates Spectral Precise Image technology with a Nano-panel Precise dual-layer detector that provides intrinsic noise reduction, combined with an AI-based reconstruction engine that allows clinicians to customize image de-noising according to their preferences.
The system also reconstructs 145 images per second, allowing for entire exams to appear in less than 30 seconds -- twice as fast as the previous generation -- and supports up to 270 exams per day, Philips said.
By capturing both high- and low-energy data in a single acquisition, Verida provides simultaneous access to conventional and spectral results without requiring separate scans or workflow changes. This enables tissue characterization and material differentiation without pre-selection or repeat acquisitions, the company said.
Verida is indicated for diagnostic imaging in radiology, interventional radiology, cardiology, and oncology treatment preparation and radiation therapy planning. It covers head, whole-body, cardiac, and vascular CT applications in patients of all ages, and is also indicated for low-dose CT lung cancer screening within established screening protocols.