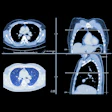
Austrian conebeam CT (CBCT) technology developer MedPhoton has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its ImagingRing CBCT system.
MedPhoton has a partnership with Mevion Medical Systems, and ImagingRing will now be available to U.S. users of that company's S250i proton therapy system, Mevion said.
The ImagingRing CBCT scanner features a 102-cm bore with ceiling rail mounting; it was integrated into Mevion's S250i system at Maastro Proton Therapy in Maastricht, the Netherlands, and has been in clinical use there since February.