Otsuka Pharmaceutical has launched its OralTag abdominal CT contrast agent in the U.S.
Developed by Otsuka subsidiary Interpharma Praha, OralTag is a dissolvable powder form of iohexol. The U.S. Food and Drug Administration (FDA) approved OralTag in March 2015 for use as a radiographic contrast agent for opacifying the gastrointestinal tract during CT scanning of the abdomen and pelvis.
Otsuka noted that OralTag is not indicated for diagnostic examination of the gastrointestinal tract and is contraindicated in patients with a known hypersensitivity to iodinated contrast agents, including iohexol.
OralTag will be marketed by imaging accessories firm Beekley Medical and commercially available from Beekley subsidiary WMB Enterprises.







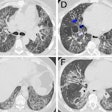






![Images show the pectoralis muscles of a healthy male individual who never smoked (age, 66 years; height, 178 cm; body mass index [BMI, calculated as weight in kilograms divided by height in meters squared], 28.4; number of cigarette pack-years, 0; forced expiratory volume in 1 second [FEV1], 97.6% predicted; FEV1: forced vital capacity [FVC] ratio, 0.71; pectoralis muscle area [PMA], 59.4 cm2; pectoralis muscle volume [PMV], 764 cm3) and a male individual with a smoking history and chronic obstructive pulmonary disorder (COPD) (age, 66 years; height, 178 cm; BMI, 27.5; number of cigarette pack-years, 43.2, FEV1, 48% predicted; FEV1:FVC, 0.56; PMA, 35 cm2; PMV, 480.8 cm3) from the Canadian Cohort Obstructive Lung Disease (i.e., CanCOLD) study. The CT image is shown in the axial plane. The PMV is automatically extracted using the developed deep learning model and overlayed onto the lungs for visual clarity.](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/03/genkin.25LqljVF0y.jpg?auto=format%2Ccompress&crop=focalpoint&fit=crop&h=112&q=70&w=112)