Radiologists should track disease distribution and distinctive dichotomous features to best identify fibrotic interstitial lung disease (ILD) patterns on CT, according to a study published March 17 in Radiology.
The results are important because accurate diagnosis of fibrotic interstitial lung disease helps determine treatment decisions and patient outcomes among a group of conditions where prognosis can be poor, noted a team led by Daniel-Costin Marinescu, MD, of the University of British Columbia in Vancouver, BC, Canada.
"Our findings will help nonexperts assign weights to the building blocks of [ILD] patterns and support refinement of future guidelines," the group wrote.
Fibrotic ILD is a group of disorders made up of various subtypes, and accurate diagnoses of these conditions rely on imaging patterns identified at high-resolution CT imaging, Marinescu and colleagues explained, noting that "clinical practice guidelines have established criteria for radiologic patterns, such as usual interstitial pneumonia (UIP) and fibrotic hypersensitivity pneumonitis (fHP), which are variably associated with common fibrotic ILD subtypes such as idiopathic pulmonary fibrosis, fHP, and connective tissue disease-associated ILD."
What's tricky is that these guidelines for recognizing radiologic patterns are based on expert opinion, and "the relative importance of various imaging features in identifying and distinguishing these patterns is unknown," the team wrote.
Marinescu's group explored the "relative weights" of imaging features that distinguish radiologic patterns identified by radiologists themselves and tracked any inconsistencies between radiologist-assigned and guideline-defined patterns. The team analyzed data from 1,498 patients who participated in the Canadian Registry for Pulmonary Fibrosis (CARE-PF); these data were analyzed between January 2021 and March 2022.
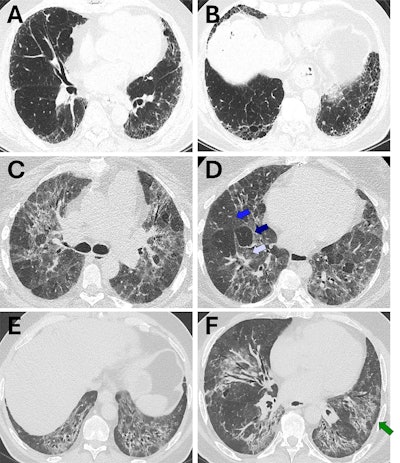 Representative cases of usual interstitial pneumonia (UIP), fibrotic hypersensitivity pneumonitis (fHP), and nonspecific interstitial pneumonia (NSIP) for which there was concordance between guideline-defined pattern (strict adherence to guidelines) and radiologist-assigned pattern (radiologist expert opinion). (A, B) Axial CT images show a case that met the criteria for guideline-defined typical UIP (2,3) and was also assigned UIP by the radiologist. The distribution is basal and peripheral, with the presence of honeycombing and reticulation and no substantial ground-glass opacity. There are no other findings that are incongruent with UIP. (C, D) Axial CT images show a case that met the criteria for guideline-defined typical fHP (2,3) and was also assigned fHP by the radiologist. The distribution is mid-upper in the craniocaudal plane and both peripheral and central in the axial plane. There is no honeycombing, but there is substantial reticulation and traction bronchiectasis. There is both lobular ground-glass opacity (white arrow in D) and hypoattenuating lung (dark blue arrow in D) alongside normal lung (blue arrow in D), forming a three-density sign. There are no other findings that are incongruent with fHP. (E, F) Axial CT images show a case that met the criteria for guideline-defined typical NSIP, defined using the 2008 American Thoracic Society workshop report (9), and was also assigned NSIP by the radiologist. The distribution is basal in the craniocaudal plane and both peripheral and central in the axial plane, with a prominent element of subpleural sparing (arrow in F). There is substantial ground-glass opacity, both pure and admixed with fibrosis. There are no other findings that are incongruent with NSIP.RSNA
Representative cases of usual interstitial pneumonia (UIP), fibrotic hypersensitivity pneumonitis (fHP), and nonspecific interstitial pneumonia (NSIP) for which there was concordance between guideline-defined pattern (strict adherence to guidelines) and radiologist-assigned pattern (radiologist expert opinion). (A, B) Axial CT images show a case that met the criteria for guideline-defined typical UIP (2,3) and was also assigned UIP by the radiologist. The distribution is basal and peripheral, with the presence of honeycombing and reticulation and no substantial ground-glass opacity. There are no other findings that are incongruent with UIP. (C, D) Axial CT images show a case that met the criteria for guideline-defined typical fHP (2,3) and was also assigned fHP by the radiologist. The distribution is mid-upper in the craniocaudal plane and both peripheral and central in the axial plane. There is no honeycombing, but there is substantial reticulation and traction bronchiectasis. There is both lobular ground-glass opacity (white arrow in D) and hypoattenuating lung (dark blue arrow in D) alongside normal lung (blue arrow in D), forming a three-density sign. There are no other findings that are incongruent with fHP. (E, F) Axial CT images show a case that met the criteria for guideline-defined typical NSIP, defined using the 2008 American Thoracic Society workshop report (9), and was also assigned NSIP by the radiologist. The distribution is basal in the craniocaudal plane and both peripheral and central in the axial plane, with a prominent element of subpleural sparing (arrow in F). There is substantial ground-glass opacity, both pure and admixed with fibrosis. There are no other findings that are incongruent with NSIP.RSNA
Radiologic patterns on CT exams were identified according to American Thoracic Society clinical guidelines (i.e., guideline-defined pattern) and radiologist impression (i.e., radiologist-assigned pattern). The group used receiver operating characteristic curves to evaluate how particular CT imaging features distinguished UIP, fHP, and NSIP patterns. Of the 1,498 patients, radiologists assigned UIP patterns to 36%, fHP patterns to 17%, NSIP patterns to 33%, and a "no confident pattern" category to 14%.
The investigators reported the following:
- More honeycombing, less total ground-glass opacity (GGO), and less pure GGO distinguished UIP from non-UIP (with area under the receiver operating characteristic curves [AUC] of 0.75, 0.81, and 0.77, respectively).
- More hypoattenuating lung differentiated fHP from non-fHP (AUC, 0.84).
- More total GGO and less honeycombing distinguished NSIP from non-NSIP (AUC, 0.71 and 0.7, respectively) -- with features with "greater than 10% lung involvement commonly demonstrating specificities of 90% or greater."
They also found that certain patterns, such as admixed GGO, prompted radiologists to disagree with guideline-defined UIP, while features such as consolidation and peripheral and basal distributions prompted them to disagree with guideline-defined fHP. Honeycombing and reticulation patterns caused radiologists to disagree with guideline-defined NSIP.
The bottom line, according to Marinescu's group, is that features most helpful for pattern identification according to experienced radiologists were related to distribution or distinctive findings, with an odds ratio of ≥ 3.
The study results are thought-provoking, but more research is needed, wrote Julianna Czum, MD, and Stacy-Ann Brown, MD, both of Johns Hopkins University in Baltimore, in an accompanying editorial.
"We remain uncertain about the adequacy of the fixed ILD patterns used in this study and recommended by current guidelines," Czum and Brown noted. "More than one distinct guideline-concurrent radiologic pattern can exist on a single CT scan, which correlates with the well-defined phenomenon of multiple histologic patterns being present within the same biopsy specimen … More nuanced, feature‑level assessments may offer a more faithful representation of disease processes and better correlate with clinical outcomes such as lung function trajectory and transplant‑free survival."
Access the full study here.