Pharmaceutical company Humanetics has acquired exclusive worldwide licenses from Henry Ford Health System in Detroit to commercially develop therapeutic agents that may provide short- and long-term postexposure protection from both lethal and nonlethal levels of ionizing radiation.
The agreement gives Humanetics of Eden Prairie, MN, the ability to develop and market two drugs, one designed for use within 24 hours of exposure and the other to be given after 24 hours of exposure. The primary role of the new treatments will be to repair damage to healthy tissues caused by radiation, as well as to prevent the onset of acute radiation syndrome (ARS) and delayed effects of acute radiation exposure (DEARE).
Researchers at Henry Ford's department of radiation oncology developed the new treatments to reduce radiation injury to normal tissue. The treatments are intended to help patients undergoing radiation therapy, in addition to those receiving CT scans. They also are being developed for individuals who may have been exposed to ionizing radiation during a terrorist act or other nuclear or radiologic event.
Efficacy studies are scheduled to begin immediately for both treatments, according to Jon Zenk, MD, chief medical officer at Humanetics.
In addition to the new licenses, Humanetics has an exclusive worldwide license to develop BIO 300, a medical radiation countermeasure drug designed to treat acute radiation syndrome, with the Armed Forces Radiobiology Research Institute in Bethesda, MD.
Related Reading
Study to test radiation protection drug, September 14, 2009
Copyright © 2010 AuntMinnie.com






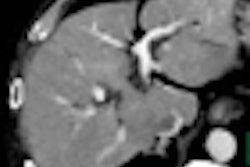
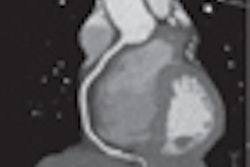


![Images show the pectoralis muscles of a healthy male individual who never smoked (age, 66 years; height, 178 cm; body mass index [BMI, calculated as weight in kilograms divided by height in meters squared], 28.4; number of cigarette pack-years, 0; forced expiratory volume in 1 second [FEV1], 97.6% predicted; FEV1: forced vital capacity [FVC] ratio, 0.71; pectoralis muscle area [PMA], 59.4 cm2; pectoralis muscle volume [PMV], 764 cm3) and a male individual with a smoking history and chronic obstructive pulmonary disorder (COPD) (age, 66 years; height, 178 cm; BMI, 27.5; number of cigarette pack-years, 43.2, FEV1, 48% predicted; FEV1:FVC, 0.56; PMA, 35 cm2; PMV, 480.8 cm3) from the Canadian Cohort Obstructive Lung Disease (i.e., CanCOLD) study. The CT image is shown in the axial plane. The PMV is automatically extracted using the developed deep learning model and overlayed onto the lungs for visual clarity.](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/03/genkin.25LqljVF0y.jpg?auto=format%2Ccompress&crop=focalpoint&fit=crop&h=112&q=70&w=112)