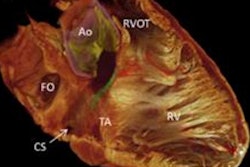
British Virgin Islands-based Cara Medical has secured clearance from the U.S. Food and Drug Administration (FDA) for its Cara System, a CT angiography-based platform for noninvasive, patient-specific 3D visualization of the cardiac conduction system.
The system is intended to support preplanning and guidance of structural heart and pacing procedures, including transcatheter aortic valve replacement and conduction system pacing, the company said. It integrates two components: the Cara Metis Simulator, a preprocedural planning algorithm that generates a personalized 3D map of the cardiac conduction system, and the Cara Atlas Navigator, an intraprocedural guidance platform that overlays the conduction system model onto live fluoroscopic images, according to the firm.
The Cara System operates on a standard workstation, integrates with existing fluoroscopy systems, and uses AI-based algorithms for CT segmentation and catheter detection, Cara Medical said. The company plans to begin its U.S. commercial launch in the coming months.






![Axial images from unenhanced calcium score cardiac CT (left) and curved planar reformation images from CT angiography (right) show that higher long-term exposure to air pollution is associated with greater coronary artery calcium and more obstructive coronary artery disease (CAD). Top row: Images in a 68-year-old male patient with higher 10-year mean ambient air pollution exposure (7.9 μg/m3 for particulate matter measuring ≤2.5 μm in diameter [PM2.5] and 17.4 parts per billion [ppb] for NO2) with extensive CAD (coronary artery calcium score [CACS] >1,000 and obstructive CAD [≥70% diameter stenosis]). Bottom row: Images in a 57-year-old female patient with lower 10-year mean ambient air pollution exposure (6.3 μg/m3 for PM2.5 and 4.6 ppb for NO2) with no CAD (CACS = 0 and no obstructive stenosis).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/06/hanneman.r6SMLzkezo.png?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)









