Contrast agents and thin-slice MDCT protocols are delivering a potent one-two punch in abdominal imaging. Investigators from the U.K. recently demonstrated the broad clinical utility of dual-phase MDCT for the investigation of hematuria. And radiologists from Ireland recently reported that the gastrointestinal hormone secretin boosted CT contrast enhancement significantly in multiphasic liver imaging.
At the genitourinary scientific sessions of the 2003 RNSA meeting in Chicago, Dr. Kirsty Anderson from the Churchill Hospital in Oxford presented a study of another promising technique: MDCT urography for the investigation of hematuria.
"CT is well established as superior to IVU investigation of renal stones and masses," Anderson said, though the modality remains largely untested for the investigation of hematuria.
The use of thin-slice MDCT heralds a major advance in urinary tract imaging, lead investigator Dr. Nigel Cowan wrote in an e-mail to AuntMinnie.com. "Patients with suspected urinary tract disease are often referred for multiple studies such as intravenous urography, ultrasound, CT, MRI, or retrograde uteropyelography," he wrote. "A single imaging test to evaluate the whole upper urinary tract has advantages in terms of both convenience and cost."
The study sought to investigate the use of MDCTU in 174 patients (62 men and 112 women ages 18-90, mean age 62). Ninety-nine patients had macroscopic hematuria and 75 had microscopic hematuria.
During the course of the study, the investigators dropped the unenhanced phase in order to reduce radiation dose, Anderson said. A split-bolus contrast technique enabled the acquisition of nephrographic and excretory-phase images simultaneously. Following the administration of 100 ml of nonionic contrast (iopamidol 300-370 mgI/ml), patients were taken off the table and exercised in order to encourage mixing of the contrast. Precisely 10 minutes later, nephrographic-phase images were acquired, then an additional 50 ml of contrast was administered, and excretory-phase images were acquired at 15 minutes after the first contrast administration, Anderson said.
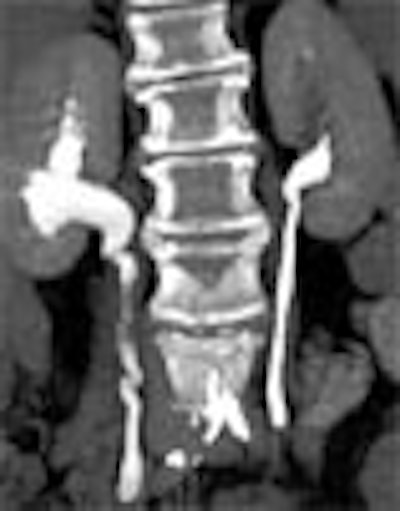
 |
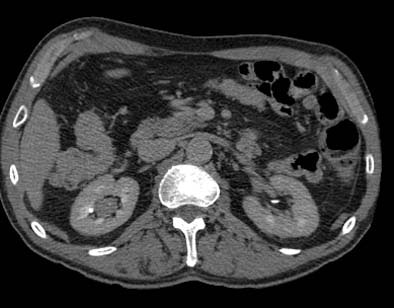
| Transitional cell cancer of the right upper ureter. Above, axial excretory-phase CT showing soft-tissue filling defect in the upper third of the ureter. Below, coronal thin-slab maximum intensity projection showing characteristic signs of ureteral transitional cell carcinoma. Images courtesy of Dr. Nigel Cowan. |
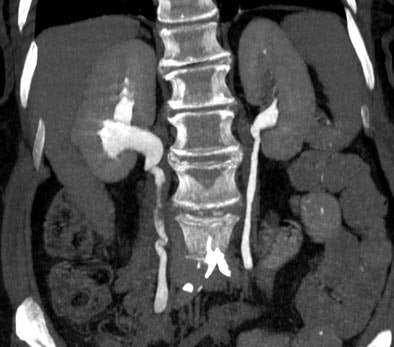 |
CT images were acquired on an eight-slice scanner using 2.5-mm slice thicknesses, 0.7- second rotation, pitch of 1.35, 120 mAs, and 120 kVp.
"Images were analyzed in all three phases in the axial plane," Anderson said. "Multiplanar (2.5-mm, 1.25-mm slice interval) reconstructions and maximum intensity projections were used when necessary." The findings were correlated with other imaging studies, cytoscopy, ureteroscopy, clinical follow-up, and biopsy and histopathology when available.
Among the 174 patients, MDCTU yielded 74 normal and 100 abnormal studies. The benign findings relevant to hematuria included 34 patients with calculi, of which 24 had renal stones, nine had ureteric stones, and one had bladder stones, Anderson said. There were also four acute myelogenous leukemia (AML) cases, one medullary sponge kidney (MSK), and one arteriovenous fistula.
MDCTU identified 53 patients with malignancies, including 10 with renal cell cancers and 43 with transitional cell cancers including renal (n=13), ureter (n=8), bladder (n=13), and multifocal (n=9). There were two false-negatives.
The malignancies included 17 renal transitional cell carcinomas, 12 of which were in the renal pelvis, two in the lower zone, and three in the upper zone. The carcinomas included 13 sessile lesions (n=8 > 10 mm; n=4 5-10 mm; n=1 < 5 mm), and four pedunculated lesions (n=2 > 10 mm; n=2 5-10 mm) according to the results.
There were 16 ureteric transitional cell carcinomas, including 13 sessile lesions (n=4 < 5 mm; n=2 5-10 mm; n=7 > 10 mm) and three cases of circumferential wall thickening < 5 mm.
MDCTU has several advantages over radiography, the researchers said, notably its ability to evaluate the entire urinary tract in a single exam. There is increased sensitivity for the detection of calculi and renal parenchymal masses as well, and the wall thickness of the entire urinary tract can be visualized. Tumors can be detected and staged in the same examination, and visualization of bladder neoplasms may also be improved.
The overall excellent results also highlighted a few potential pitfalls of MDCTU, Cowan wrote. The technique is new, so the methods have not yet been standardized. Workstation review can be time-consuming. In some cases, reformatting of CT images can mask tumors.
And regardless of the technique timing of the contrast, the unopacified ureter remained a frequent problem, as it is during IVU. Limited mixing of contrast could be seen in some cases. Barium artifact, incomplete bladder filling, and iodine allergy that prevented contrast administration caused additional suboptimal studies.
"Ureteral kinks may produce focal areas of narrowing or thickening, thereby mimicking ureteral strictures or even tiny neoplasms," Cowan wrote. Partial-volume effects also complicated the diagnosis at times, Anderson added.
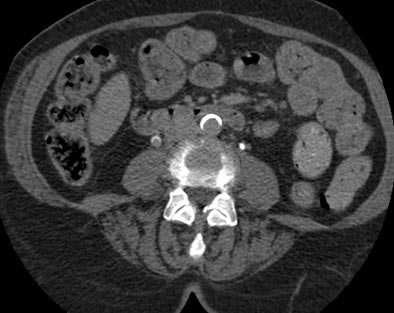
 |
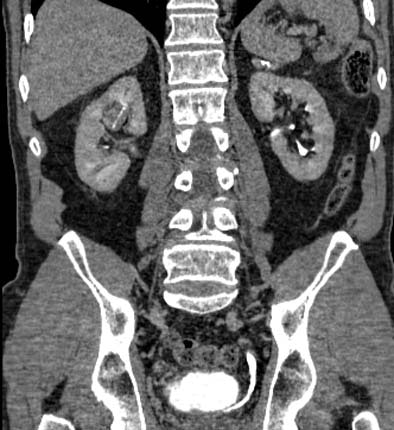
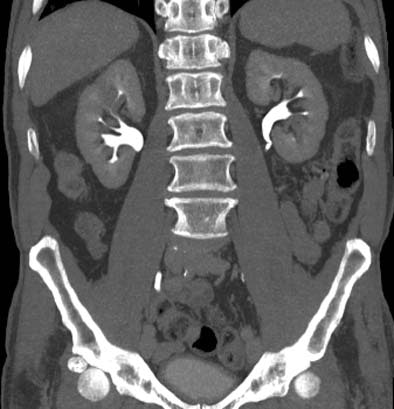
| Postprocessing or reformatting images may obscure the diagnosis. TCC clearly seen on axial CT (above) and on coronal multiplanar reformat below. But even a thin-slab MIP reformat masks detection of the right-upper-pole TCC (bottom). Images courtesy of Dr. Nigel Cowan. |
 |
 |
Finally, the radiation dose remains worrisome. Triphasic imaging, which the group abandoned early on in favor of a biphasic protocol, yielded an average radiation dose of 13.7 mSv compared to 2.5 mSv for the average IVU exam, Anderson said. The group's technique of choice, a split-bolus two-phase study, dropped the dose a bit to 11.6 mSv. Efforts are underway to reduce it further, Anderson said.
Still, studies to date have shown MDCTU to be more accurate for the investigation of hematuria than both IVU and retrograde pyelography, an imaging exam assumed to be superior to IVU for evaluating the renal collecting systems and ureters, Cowan wrote.
MDCTU is a promising technique for the investigation of hematuria, Anderson said, adding that the technique may be ready for a broader clinical role in the evaluation of urothelial pathology.
Secretin boosts hepatic enhancement
Another contrast CT study presented at the RNSA meeting demonstrated the ability of the gastrointestinal hormone secretin to boost the effectiveness of iodinated contrast in the liver.
"In a previous study we performed secretin-assisted CT of the pancreas," said Dr. Anne Marie O'Connell from the Beaumont Hospital in Dublin, Ireland. "We administered secretin three minutes prior to contrast-enhanced CT. We demonstrated increased pancreatic enhancement, and increased tumor conspicuity. We also noted that there was increased enhancement of the portal venous system."
This finding led the group to wonder if secretin might be able to boost enhancement and improve the conspicuity of hypervascular lesions in the liver as well. To find out, the group performed triphasic CT in 31 patients (mean age 66.1) with malignancy on successive days.
First, unenhanced images were acquired, followed by a pancreatic phase 40 seconds after administration of iopamidol at 5 mLs/sec, and a portal-venous phase at 70 seconds. The first day's images were acquired without secretin; on the second day, 100 IU of secretin was administered two, three, and four minutes prior to contrast administration and CT imaging.
"CT enhancement was measured at three locations of the liver in the unenhanced phase, the pancreatic, and the portal venous phase," O'Connell said. "We also looked at blood vessels; we measured the attenuation of the celiac artery, the superior mesenteric vein, the hepatic artery, and the portal vein."
Celiac artery enhancement was 293.70 HU before secretin and 274.9 HU after, an overall reduction of 18.8 HU. Hepatic-artery enhancement was 274.9 HU before secretin and 270.55 after -- representing a decrease in enhancement of 18.8 HU for the celiac artery (p<0.001) and a slight increase of 0.2 for the hepatic artery post-secretin. The most important increases were in portal-vein enhancement: 214.15 HU before secretin and 235.75 HU after, an overall increase of 21.6 HU (P<0.0079).
In the pancreatic phase, "the portal vein demonstrated a 38% increase in attenuation after the administration of secretin: this is statistically significant," O'Connell said, adding that the increased pancreatic-phase enhancement after secretin administration is due to increased contrast-medium delivery into the vein.
"We know that secretin increases contrast-medium delivery by increasing portal blood flow," she said. "We have also shown that it accelerates the timing of the delivery of the (contrast)," making the portal-venous phase occur earlier.
"We have demonstrated increased liver enhancement in the pancreatic phase due to increased contrast-medium delivery in the portal vein," O'Connell said. "The pancreatic phase and the portal-venous phase occur earlier when secretin is administered. Therefore, we may have to...reduce our timing delays." Further work is on the way, she said, including studies to determine the optimal timing and protocols. Meanwhile, the three-minute delay between secretin and contrast administration seems to work best, she said.
One problem with secretin is its high cost, about 100 euros a dose in Europe and as much as $300-$500 in the U.S., O'Connell said, adding that it's definitely worth the cost, since the group has found malignancies that were not detected without the agent.
By Eric BarnesAuntMinnie.com staff writer
March 1, 2004
Related Reading
Spiral CT protocol predicts kidney stone composition, May 19, 2003
CT better than IVP in diagnosing acute urolithiasis, September 11, 2002
Lowering CT dose yields usable results in renal colic, May 24, 2002
Slashing CT dose in clinical practice, July 25, 2001
Copyright © 2004 AuntMinnie.com