X-ray dark-field chest imaging -- a new technique touted as the most significant advance in standard chest x-ray in 100 years -- has shown for the first time it may help diagnose lung disease in humans, according to a study published August 24 in Radiology.
German researchers designed and built a prototype and tested the system in healthy patients. They confirmed x-ray dark-field chest imaging picks up signals in the lungs that are undetected in standard chest x-rays and established its qualitative and quantitative characteristics -- a crucial step toward evaluating the new system in future trials.
"These findings prove that the dark-field signal is indeed sensitive to the subject's lung condition alone and is independent from demographic factors, highlighting its potential value for diagnosis and monitoring of respiratory diseases," wrote a team led by first author Dr. Florian Gassert of the Technical University of Munich.
The researchers first investigated x-ray dark-field chest imaging in 2008. In contrast to attenuation-based conventional radiography, dark-field imaging harnesses the wave properties of x-rays. Specifically, the system detects the signal of ultra-small-angle scattering that takes place in water-to-air transitions in the alveolar structure of the lungs.
In attenuation-based radiography, dense structures generate a high signal, while in dark-field imaging, the small-angle scattering in lung tissue generates a high signal. Because dark-field x-ray imaging excludes unscattered photons, the space around the lungs appears dark (because there is no material there to scatter photons).
Since they developed the system, the researchers have conducted a number of animal studies that show the signal in x-ray dark-field chest imaging decreases in lung diseases that interfere with alveolar structure, such as emphysema, fibrosis, lung cancer, and ventilation-induced lung damage.
In the current study, they sought to describe the qualitative and quantitative characteristics of x-ray dark-field images for the first time in humans.
Between October 2018 and January 2020, the researchers enlisted 40 healthy patients who underwent chest CT as part of their diagnostic workup. Inclusion criteria were a normal chest CT scan, the ability to consent, and the ability to stand upright without help.
Importantly, the researchers developed a prototype that acquired both attenuation-based and dark-field chest radiographs simultaneously. Each patient's total dark-field signal was correlated with his or her lung volume, and the dark-field coefficient was correlated with age, sex, weight, and height.
The researchers found normal human lungs on dark-field chest x-ray imaging produced high signal, while the surrounding osseous structures had low signal and soft tissue produced no signal. The average total dark-field signal intensity over all participants for the entire lung was (17 ± 4) × 10-3 m2 and the total signal demonstrated a positive correlation with lung volume (r = 0.61, p < 0.001).
In addition, no differences were found between men and women and age, weight, and height did not influence the dark-field signal.
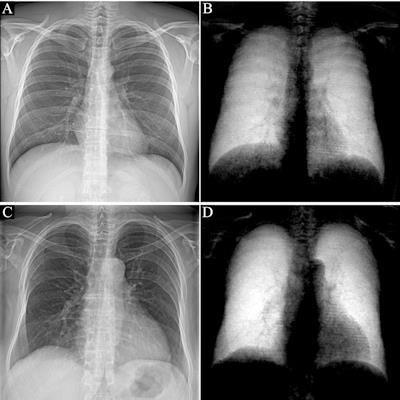
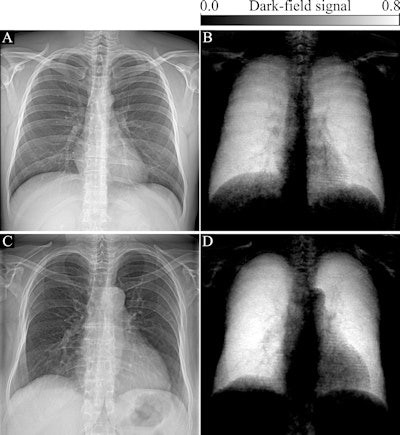 Attenuation-based (left) and dark-field (right) posteroanterior radiographs of the thorax in example subjects. Images were obtained in (A, B) a 33-year-old man and (C, D) an 81-year-old man. The same window and level settings were applied within the respective modality. While attenuation-based images show attenuating structures, such as the bones and the soft tissue, dark-field images show the amount of tissue interfaces in the beam path. Image courtesy of Radiology.
Attenuation-based (left) and dark-field (right) posteroanterior radiographs of the thorax in example subjects. Images were obtained in (A, B) a 33-year-old man and (C, D) an 81-year-old man. The same window and level settings were applied within the respective modality. While attenuation-based images show attenuating structures, such as the bones and the soft tissue, dark-field images show the amount of tissue interfaces in the beam path. Image courtesy of Radiology."Because of the nature of signal generation in dark-field imaging, bone structures and soft tissue generate only a minimal dark-field signal compared with lung tissue. This feature allows for a detailed depiction of lung tissue without impairment by surrounding structures. We found that the quantitative x-ray dark-field coefficient based on the total dark-field signal and lung size is independent from the subject's characteristics," the researchers wrote.
The team noted limitations, namely that only healthy subjects were included. However, future studies will assess abnormal changes in the lung tissue, they concluded.
In an accompanying editorial, Dr. Hiroto Hatabu, PhD, and Bruno Madore, PhD, of Harvard Medical School, wrote that dark-field x-ray is still in its infancy -- even results in animal models are still very preliminary -- but that the work is an important step toward establishing normative values for dark-field x-ray chest imaging in humans.
Chest imaging has come a long way since it first emerged as a clinical test after the discovery of x-rays by Wilhelm C. Röntgen, yet through it, all x-ray imaging has continued to only exploit the particle aspect of x-ray photons rather than their wave aspects, they wrote.
"We welcome the addition of the dark-field approach to the universe of chest radiography methods available for human imaging and diagnoses," Hatabu and Madore wrote.