Takotsubo cardiomyopathy or Transient Left Ventricular Apical Ballooning Syndrome:
Clinical:
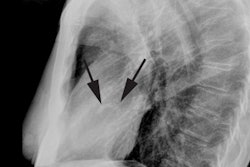
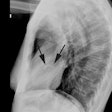
Transient LV apical ballooning syndrome is a rare form of transient LV systolic dysfunction involving the apical and/or mid segments (accompanied by ballooning of the involved segments) that mimics an acute coronary syndrome/acute MI [1,3,7]. The name Takotsubo is related to the morphology of the left ventricle, which shows a resemblance to a Japanese round-bottomed narrow-necked fishing pot used for trapping octopus [4]. Patients present with anginal, substernal chest pain, ST segment elevations on ECG (in the precordial leads in up to 68% of patients [3]), diffuse T-wave inversions (up to 97% of cases [3]), and often mild elevated cardiac enzymes (86% of patients have mildly elevated Troponin levels and 74% have slightly elevated creatine kinase-MB levels) [2,3]. In contrast to the pattern associated with acute MI, the cardiac enzymes in Takotsubo cardiomyopathy (TC) peak at presentation and quickly normalize [3]. The disorder is characterized by extensive regional wall motion abnormalities with hypokinesis or dyskinesis of the apical segments and hypercontractility of the basal segments in the absence of atherosclerotic disease at catheterization (significant CAD is found in only 5% of patients at angiography) [1,2,3]. The acute phase of the disorder is felt to be related to metabolic dysfunction at the cellular level [1]. Formal diagnostic criteria have not yet been established, but the following have been suggested: 1- transient LV wall motion abnormalities involving the apical and/or midventricular myocardial segments with wall motion abnormalities extending beyond a single epicardial coronary distribution; 2- absence of obstructive CAD; and 3- new ECG abnormalities such as transient ST-segment elevation and/or diffuse T-wave inversions or Troponin T evelation [4].
The disorder is most frequently seen in post menopausal women-
87%
of reported cases (average age 62-75; mean age 68 years), often in
the
setting of acute emotional or physiologic stress [2,3,4]. Although
the
exact pathophysiology of the disorder is unknown, there is
evidence
that excessive sympathetic stimulation is integral to the
development
of TC (physical or emotional stress is the precipitating factor in
about 80% of cases) and neurogenic myocardial stunning has been
demonstrated in patients using I-123 MIBG scintigraphy [3,4].
Stress
induced coronary epicardial spasm or endothelial dysfunction
resulting
in myocardial stunning is another possible etiology for the
condition
[4]. In the acute phase, the patient is commonly treated with
medication for systolic heart failure- beta-blockers,
ACE-inhibitors, and diuretics [7].
Most patients fully recover within a few weeks (in-hospital mortality is between less than 1% to 4%) [2,3,7]. In hopsital mortality is higher in males compared to females (up to 8%) [7]. However, patients can develop severe LV dysfunction requiring supportive measures- mid and distal LV akinesia with compensatory basal hyperkinesia can cause LV cavity obliteration that results in LV outflow obstruction and intraventricular pressure gradients [3]. There is evidence that dobutamine is contraindicated in patients with TC, especially in the presence of outflow obstruction and that beta blockade may be beneficial in the treatment of the disorder [3]. The recurrence of the disorder is rare (estimated at 2.9%/year) [4].
A variant presenting as basal ballooning (that spares the mid-LV region and has paradoxical apical hyperkinesis) has recently been described in young pre-menopausal women [3].
X-ray:
Stress myocardial perfusion imaging during the acute phase will demonstrate a reversible perfusion defect in the apex and reduced apical coronary flow reserve [1,4]. Metabolic imaging with FDG has been reported to demonstrate a severe reduction in glucose metabolism in the apical segments (during hyperinsulinemic-euglycemic clamp imaging) [1]. A flow-metabolism mis-match pattern has been described with relatively preserved or moderately reduced myocardial perfusion to the apex, but markedly abnormal decreased metabolism (with either BMIPP or FDG) [3,5]. A reversible impairment of coronary flow reserve in the apical myocardial segements using quamtitative perfusion PET has been demonstrated suggesting that microcirculation dysfunction plays a key role in this disorder [5]. In general, the regional reduction of perfusion and impairment in glucose metabolism have a tendency to resolve completely within 3 months from presentation [4].
Regional denervation has also been reported in the apex during the acute phase on I123 MIBG imaging with decreased apical tracer uptake [1,5,7]. Normalization of FDG uptake with persistent decreased apical I123 MIBG uptake can be seen even at one year followup [5].
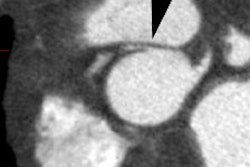
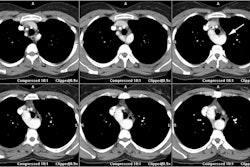
On MR, there is characteristic apical ballooning with akinesis or severe hypokinesis, while the basal portions of the LV demonstrate hyperkinesis [2,6]. Global edema with high signal intensity in the mid and apical myocardium suggests the diagnosis (patchy signal would be more suggestive of myocarditis) [7]. Delayed contrast enhanced MR imaging does NOT show any evidence of delayed enhancement on post gadolinium imaging [1]. The apical abnormality resolves and normal function and imaging exams can be seen by 3 months following the event [1].
REFERENCES:
(1) J Nucl Cardiol 2008; Feola M, et al. Reversible impairment of coronary flow reserve in takotsubo cardiomyopathy: a myocardial PET study. 15: 811-817
(2) Radiographics 2009; Cummings KW, et al. A pattern-based approach to assessment of delayed enhancement in nonischemic cardiomyopathy at MR imaging. 29: 89-103
(3) J Nucl Cardiol 2009; Dorfman TA, Iskandrian AE. Takotsubo cardiomyopathy: state-of-the-art review. 16: 122-134
(4) J Nucl Cardiol 2009; Verberne HJ, et al. Persisting myocardial sympathetic dysfunction in takotsubo cardiomyopathy. 16: 321-324
(5) J Nucl Cardiol 2009; Galiuto L, et al. Non-invasive imaging
of
microvascular damage. 16: 811-831
(6) Radiology 2012; O'Donnell DH, et al. Cardiac MR imaging of
nonischemic
cardiomyopathies: imaging protocols and spectra of appearances.
262:
403-422
(7) J Nucl Cardiol 2014; Verschure DO, et al. Tako-tsubo
cardiomyopathy: how to understand possible pathophysiological
mechanism and the role of 123I-MIBG imaging. 21:
730-738